VK2735 for Weight Loss: Oral GLP-1/GIP Results and Current Status
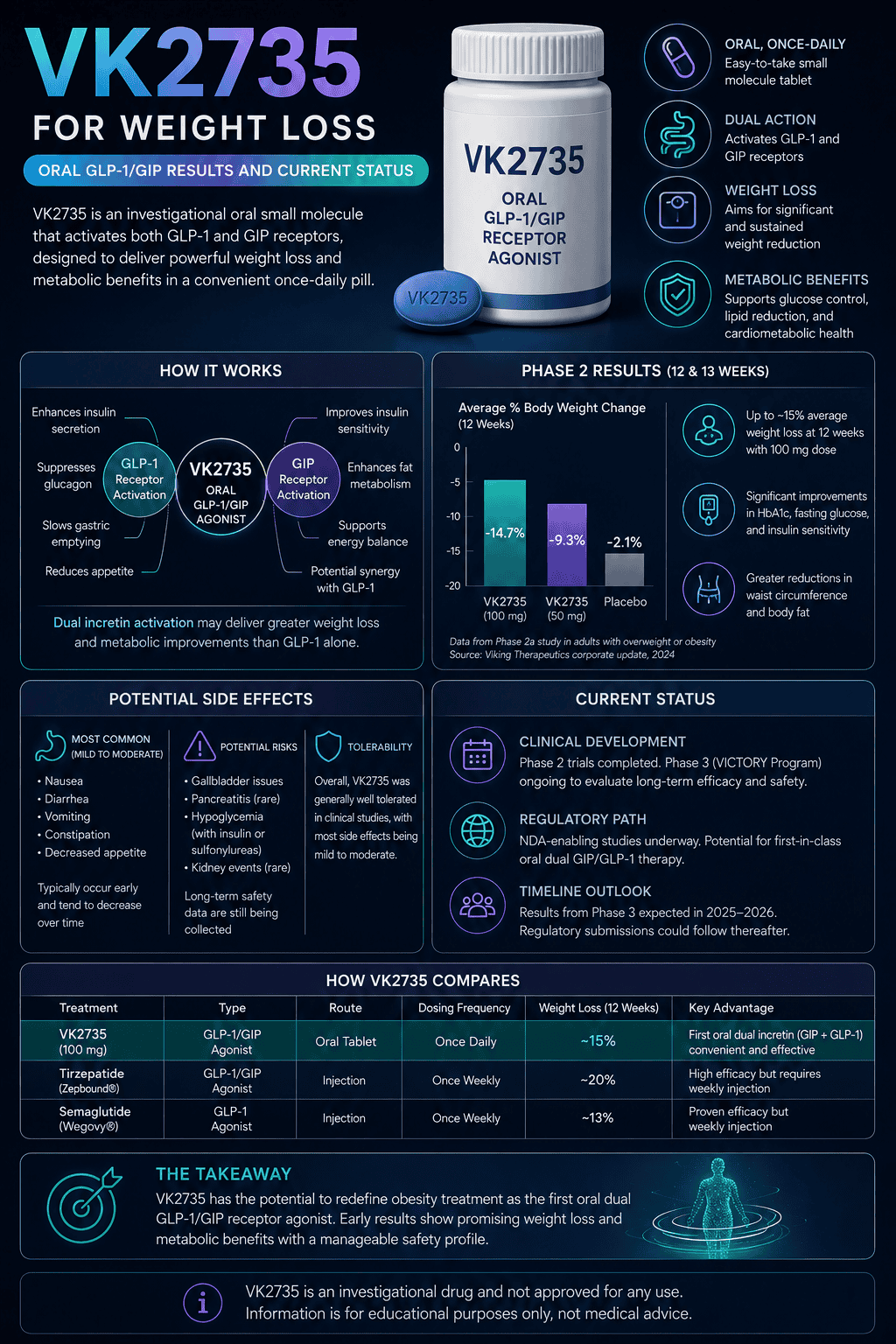
VK2735 is an investigational dual GLP-1/GIP receptor agonist being studied for weight loss in both oral and injectable forms. The oral tablet has shown meaningful short-term weight loss in Phase 2 data, but it is not FDA-approved and does not yet have long-term Phase 3 results.
In a 13-week Phase 2 oral trial, once-daily VK2735 produced up to 12.2% mean body weight reduction from baseline, compared with 1.3% for placebo. The main caution is tolerability: gastrointestinal side effects and treatment discontinuations remain important to watch.
This article focuses on the oral VK2735 tablet, with subcutaneous VK2735 included only where it helps explain the broader development program.
Who this is for: Readers tracking oral GLP-1/GIP weight-loss drugs, clinical trial results, and VK2735’s development status.
Who this is not for: Anyone trying to buy, compound, or use VK2735 outside a regulated clinical trial.
Reviewed by VerifiedSupps Editorial Team • Last reviewed: May 12, 2026
The answer-first view: VK2735 looks like one of the more notable oral incretin drug candidates for obesity, but the evidence is still early. The oral tablet has encouraging 13-week data, while the injectable version is further along in Phase 3 studies.
The most useful way to read the results is with balance. A 12.2% average weight reduction over 13 weeks is substantial for a short trial, but it does not prove long-term durability, cardiovascular benefit, real-world adherence, or final safety.
VK2735 should also not be treated like a supplement. It is a clinical-stage drug candidate, closer in category to prescription obesity medicines than to nutrition support products. For broader context on emerging obesity drugs, see our guides to retatrutide weight loss data and status and CagriSema for weight loss.
Key Takeaways
- VK2735 is a dual GLP-1/GIP receptor agonist being developed by Viking Therapeutics in oral and subcutaneous forms.
- Oral VK2735 Phase 2 results showed up to 12.2% mean weight loss from baseline after 13 weeks, with dose-dependent effects.
- The oral trial reported no clear weight-loss plateau at 13 weeks, but the study was short and not designed to prove long-term maintenance.
- Gastrointestinal side effects were common. Overall treatment discontinuation was 28% with VK2735 versus 18% with placebo in the Phase 2 oral study.
- Oral VK2735 is not approved or commercially available. Viking’s latest update points to oral Phase 3 initiation expected in late 2026.
- Any VK2735 use outside a clinical trial should be avoided.
Does VK2735 work for weight loss?
Early clinical data suggest VK2735 can produce clinically meaningful weight loss, but the oral formulation still needs longer Phase 3 trials. The best current evidence for oral VK2735 is a 13-week Phase 2 dose-finding study.
VK2735 is designed to activate both GLP-1 and GIP receptors. That places it in the same broad dual-incretin category as tirzepatide, though VK2735 remains investigational and should not be assumed to have the same final efficacy, safety, dosing, or approval status.
Mechanism
- GLP-1 receptor activation is associated with reduced appetite, improved satiety, slower gastric emptying, and better glucose regulation.
- GIP receptor activation may add metabolic and appetite-related effects when paired with GLP-1 activity, but the exact contribution can vary by drug.
- The real-world outcome is not “fat burning” in isolation; weight loss is mainly expected through lower energy intake and improved appetite control.
- Mechanism does not prove durability. Long-term weight maintenance, lean mass effects, and safety require longer controlled studies.
For readers comparing multi-receptor obesity drugs, our guide on how retatrutide works explains a related but distinct multi-agonist strategy. VK2735 is a dual GLP-1/GIP agonist; retatrutide is being studied as a triple agonist.
How much weight did people lose with oral VK2735?
In the 13-week oral Phase 2 study, the highest VK2735 dose group lost an average of 12.2% of body weight from baseline, compared with 1.3% for placebo. Weight loss was dose-dependent and continued through week 13 without a reported plateau.
| Study arm | Mean weight change at 13 weeks | Placebo-adjusted change | Participants with ≥10% weight loss |
|---|---|---|---|
| Placebo | -1.3% | Reference | 5% |
| VK2735 15 mg | -2.3% | -1.0% | 8% |
| VK2735 30 mg | -7.0% | -5.7% | 35% |
| VK2735 60 mg | -8.7% | -7.4% | 40% |
| VK2735 90 mg | -11.1% | -9.8% | 59% |
| VK2735 120 mg | -12.2% | -10.9% | 80% |
The same trial reported that up to 97% of VK2735-treated participants achieved at least 5% weight loss, compared with 10% for placebo. Those are encouraging responder data, but they come from a controlled short-term trial and should not be interpreted as typical real-world outcomes.
Viking also reported an exploratory maintenance cohort in which participants were titrated to 90 mg daily and then reduced to 30 mg daily. Weight loss appeared to continue or be maintained after the dose reduction, but this was exploratory and needs confirmation before drawing firm conclusions.
A separate subcutaneous VK2735 Phase 2 study reported up to 14.7% mean weight loss after 13 weekly doses. That supports the broader VK2735 program, but oral and injectable results should not be compared as if they were tested head-to-head.
What oral VK2735 dose was used in trials?
Oral VK2735 has no approved dose. The Phase 2 oral trial studied once-daily doses of 15, 30, 60, 90, and 120 mg, with titration used in the higher-dose groups.
This matters because trial dosing is not the same as patient dosing. A Phase 3 program may use different starting doses, titration steps, maintenance doses, or eligibility rules depending on safety and regulatory feedback.
- Lower-dose arms: 15 mg and 30 mg were studied as daily doses for 13 weeks.
- Higher-dose arms: 60, 90, and 120 mg arms used gradual dose escalation.
- Phase 1 oral data: Earlier 28-day testing included doses up to 100 mg daily and reported up to 8.2% mean weight loss from baseline.
- Maintenance question: An exploratory arm suggested lower-dose oral maintenance may be possible, but this is not established.
The practical takeaway is simple: there is no safe consumer dosing protocol for VK2735. Anyone seeing VK2735 sold online should treat that as a major red flag. For broader context, read our science-based overview on whether peptides are safe.
When could VK2735 be approved or available?
VK2735 is not approved for weight loss as of May 12, 2026. Viking’s most recent corporate update said oral VK2735 Phase 3 initiation is expected in the fourth quarter of 2026.
The injectable version is further along. Viking’s VANQUISH Phase 3 program for subcutaneous VK2735 includes two 78-week trials: one in adults with obesity or overweight with weight-related comorbidity, and one in adults with obesity and type 2 diabetes. Viking reported that both trials were fully enrolled in the first quarter of 2026.
That does not mean approval is imminent. Phase 3 trials must be completed, analyzed, submitted to regulators, and reviewed. The final label, dose, warnings, and eligible population are still unknown.
- Oral VK2735: Phase 2 complete; Phase 3 expected to begin later in 2026.
- Subcutaneous VK2735: Phase 3 VANQUISH trials ongoing and fully enrolled.
- Maintenance dosing: Separate study ongoing, with data expected in 2026.
- Commercial availability: Not available unless and until regulatory approval is granted.
What are the side effects of VK2735?
The most common VK2735 side effects reported so far are gastrointestinal, including nausea, vomiting, diarrhea, constipation, and abdominal discomfort. Most reported events were mild or moderate, but discontinuations were not trivial in the oral Phase 2 trial.
In the 13-week oral study, adverse-event discontinuation occurred in 20% of VK2735-treated participants versus 13% with placebo. Overall treatment discontinuation was 28% with VK2735 versus 18% with placebo, and the highest-dose group had a 38% early treatment discontinuation rate.
- Nausea: Reported in 58% of VK2735-treated participants versus 48% with placebo.
- Vomiting: Reported in 26% of VK2735-treated participants versus 10% with placebo.
- GI timing: Events were generally reported early in treatment and decreased with repeat dosing.
- Severity: Viking reported that 99% of GI-related treatment-emergent adverse events were mild or moderate.
Those data are encouraging in some respects, but they do not remove the need for caution. A 13-week trial cannot fully answer questions about gallbladder events, pancreatitis risk, dehydration risk, kidney complications, nutritional adequacy, lean mass loss, or rare adverse events.
Rapid weight loss can also reduce lean mass if nutrition and resistance training are neglected. For related context, see our article on retatrutide and muscle loss and our guide to how much protein you actually need.
Who should avoid VK2735 or be especially cautious?
Outside a registered clinical trial, everyone should avoid VK2735. It is investigational, and there is no approved product, dose, prescribing label, or consumer safety framework.
If VK2735 eventually reaches prescribing review, caution will likely be especially important for people who overlap with known GLP-1 and dual-incretin risk groups. The final contraindications would depend on the approved label, if one is granted.
- People who are pregnant, trying to become pregnant, or breastfeeding.
- People with a history of pancreatitis, gallbladder disease, or severe gastrointestinal motility disorders.
- People using insulin or sulfonylureas, where appetite changes and glucose effects may increase hypoglycemia risk.
- People with kidney disease or a history of dehydration from vomiting or diarrhea.
- People with active eating disorders or high risk of under-eating.
- People considering gray-market peptides, research vials, or compounded products labeled as VK2735.
The most conservative interpretation is that VK2735 belongs in the clinical trial setting until safety, dosing, and regulatory review are complete.
How does VK2735 compare with other weight-loss drug candidates?
VK2735 is notable because Viking is developing both oral and injectable versions of the same dual GLP-1/GIP agonist. That could be useful if future data support switching from injection to tablet maintenance, but this remains an unproven clinical strategy.
Compared with approved therapies, VK2735 has less evidence. Approved drugs have completed larger, longer registration trials and have prescribing labels. VK2735’s oral data are promising but still short-term.
Compared with other pipeline candidates, VK2735 sits in a crowded and fast-moving obesity research landscape. Readers may also want to compare:
- Retatrutide vs tirzepatide for dual versus triple incretin comparisons.
- Retatrutide vs semaglutide for weight loss for GLP-1 versus multi-agonist context.
- Cagrilintide for weight loss and amycretin for weight loss for amylin-based approaches.
- MariTide for weight loss, mazdutide for weight loss, and survodutide for weight loss for other incretin-pathway candidates.
- Pemvidutide for weight loss and fatty liver for a related metabolic drug-development angle.
The careful takeaway: VK2735 is promising, especially because of the oral tablet data, but it is too early to call it superior to approved drugs or other late-stage candidates.
References
- Viking Therapeutics. Phase 2 VENTURE-Oral Dosing Trial Data Presented at ECO 2026.
This source supports the updated May 2026 oral VK2735 results, including the 12.2% mean body weight reduction, responder rates, dose response, and no-plateau language through week 13.
- Viking Therapeutics. Positive Top-Line Results from Phase 2 VENTURE-Oral Dosing Trial of VK2735.
This release provides the detailed oral Phase 2 dose table, placebo-adjusted weight changes, discontinuation rates, and common gastrointestinal adverse events used in this article.
- Viking Therapeutics. First Quarter 2026 Financial Results and Corporate Update.
This source supports the current development status, including fully enrolled subcutaneous VANQUISH Phase 3 studies, expected oral Phase 3 initiation in late 2026, and ongoing maintenance dosing research.
- ClinicalTrials.gov. NCT06828055: VK2735 for Weight Management Phase 2.
This trial registry supports the study design: randomized, double-blind, placebo-controlled, 13-week oral VK2735 dosing in adults with obesity or overweight with at least one weight-related comorbidity.
- Bays HE, Toth P, Alkhouri N, et al. Weekly Subcutaneous VK2735, a GIP/GLP-1 Receptor Dual Agonist, for Weight Management. Obesity. 2026.
This peer-reviewed Phase 2 publication supports the broader VK2735 program and the reported weight-loss effects of the subcutaneous formulation over 13 weeks.
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide Once Weekly for the Treatment of Obesity. New England Journal of Medicine. 2022.
This pivotal trial supports background context for dual GLP-1/GIP receptor agonism in obesity treatment, while VK2735 remains a separate investigational drug.
- Ismaiel A, et al. Gastrointestinal adverse events associated with GLP-1 receptor agonists in adults with overweight or obesity. International Journal of Obesity. 2025.
This review supports the broader point that nausea, vomiting, diarrhea, and constipation are common adverse events across GLP-1-based obesity therapies.
FAQ
Is VK2735 available now?
No. VK2735 is investigational and is not approved for weight loss. Access should be limited to properly regulated clinical trials.
Is VK2735 a supplement?
No. VK2735 is a drug candidate, not a dietary supplement. Products marketed online as VK2735 should not be treated as safe or legitimate consumer products.
Is oral VK2735 the same as injectable VK2735?
They are being developed as different formulations of the same active therapeutic candidate. However, oral and injectable formulations can differ in dosing, absorption, tolerability, and final clinical use.
How fast did oral VK2735 start working?
Viking reported statistically significant differences versus placebo beginning at week 1 for oral doses above 15 mg. That early response is interesting, but long-term outcomes matter more than week-1 changes.
Does VK2735 cause nausea?
Nausea was common in the oral Phase 2 trial. Most nausea was mild or moderate, but gastrointestinal side effects were also the most common reason participants stopped treatment.
Is VK2735 better than tirzepatide or semaglutide?
That cannot be concluded from current data. VK2735 has promising early results, but it has not completed the same depth of long-term evidence or regulatory review as approved obesity medications.
Medical Disclaimer
This article is for educational purposes only and does not provide medical advice, diagnosis, or treatment. VK2735 is an investigational drug candidate and should not be purchased, compounded, prescribed, or used outside a regulated clinical trial unless it receives regulatory approval and is prescribed by a qualified clinician. People considering obesity treatment should speak with a licensed healthcare professional about approved options, contraindications, monitoring, nutrition, muscle preservation, and long-term weight-management planning.