By VerifiedSupps Editorial Team
Mazdutide for Weight Loss: Results, Side Effects, and How It Works
Mazdutide is a once-weekly GLP-1/glucagon dual receptor agonist that has shown meaningful weight-loss results in Chinese clinical trials. It is already approved in China for chronic weight management and type 2 diabetes, but it is not a globally available or FDA-approved weight-loss medication.
The practical answer is balanced: mazdutide is clinically serious, especially at 4 mg, 6 mg, and emerging 9 mg data, but comparisons with semaglutide, tirzepatide, and newer multi-agonists need careful trial-context reading.
This guide focuses on mazdutide weight-loss results, studied dose ranges, side effects, mechanism, liver-fat signals, and current availability; it does not provide injection instructions, sourcing advice, or a substitute for clinician-guided obesity care.
Key terms: mazdutide, IBI362, LY3305677, Xinermei, GLP-1 receptor, glucagon receptor, oxyntomodulin analogue, GLORY-1, GLORY-2
Quick Take
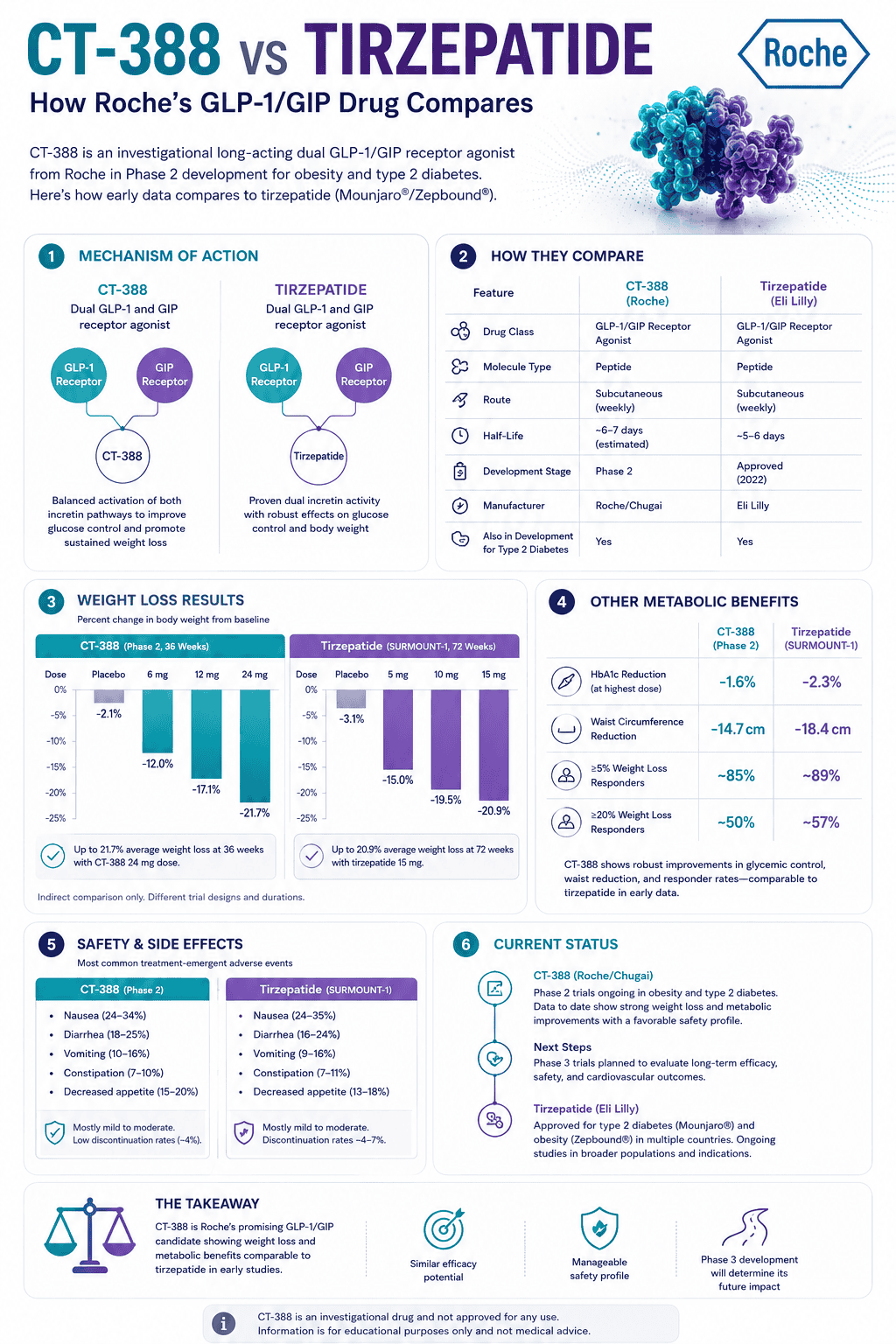
Mazdutide has strong China-based evidence: GLORY-1 showed up to 14.84% mean weight loss at 48 weeks with 6 mg, while GLORY-2 reported 18.55% mean loss at 60 weeks with 9 mg in adults with obesity. Its main side effects are gastrointestinal, and the glucagon component is the reason liver-fat and metabolic-health outcomes are part of the story.
TL;DR decision
Mazdutide is worth watching and, in China, it is already a regulated treatment option for eligible adults. Outside approved markets, do not treat online mazdutide products as safe substitutes; use approved, clinician-supervised weight-loss options instead.
Evidence standard: human trials, dose ranges, guideline-level sources when available
Who this is for: readers comparing mazdutide with GLP-1 drugs, dual agonists, retatrutide-style multi-agonists, and emerging obesity medications
Who this is not for: anyone looking for research-vial sourcing, self-injection instructions, or medical advice outside a licensed prescribing pathway
Reviewed by: VerifiedSupps Editorial Team
Last reviewed: May 12, 2026
Parent Hub
VerifiedSupps Articles Library
Use the main article hub to compare evidence-based guides on peptides, obesity medications, metabolic health, and supplement safety in one place.
Mazdutide decision decoder
Use this quick table to separate the headline results from the practical decision.
| What you want to know | Best read | What it means | Practical action |
|---|---|---|---|
| “Does mazdutide work for weight loss?” | Yes, in Chinese trials; up to 14.84% at 48 weeks with 6 mg and 18.55% at 60 weeks with 9 mg in separate Phase 3 studies | Strong evidence, but population and dose context matter | Compare it against approved options available in your country |
| “Can I use it now?” | Approved in China for specified indications; not FDA-approved or broadly available globally | Access is country-specific and prescription-based | Do not substitute research-vial products for regulated care |
| “Is it mainly a GLP-1 drug?” | No, it targets GLP-1 and glucagon receptors | The glucagon side is why liver fat and energy-expenditure claims appear | Judge outcomes, not just mechanism |
| “Is it safer than semaglutide or tirzepatide?” | No simple answer; gastrointestinal side effects are still the dominant pattern | Different receptor targets do not remove the need for medical screening | Discuss risk, history, and availability with a clinician |
Best next step (today): if you are outside China, compare mazdutide as an evidence topic, not as a self-use option.
Does mazdutide work for weight loss?
Mazdutide does appear to work for weight loss in controlled human trials, especially in Chinese adults with overweight or obesity. The cleanest interpretation is that it is a real obesity medication in approved markets, but not a universal replacement for semaglutide, tirzepatide, or clinician-guided care.
Mechanism
- GLP-1 receptor activation can reduce appetite, improve fullness, slow gastric emptying, and support glucose control.
- Glucagon receptor activation is being used to target energy expenditure and hepatic fat metabolism.
- Real-world outcomes still depend on dose, titration, tolerability, adherence, nutrition quality, resistance training, and medical eligibility.
What were the mazdutide trial results?
The strongest obesity evidence comes from GLORY-1 and GLORY-2. GLORY-1 supports the approved lower-dose weight-management indication in China, while GLORY-2 supports the higher-dose 9 mg strategy for adults with more severe obesity.
| Evidence layer | Population | Weight-loss result | Clean interpretation |
|---|---|---|---|
| Phase 2 dose-finding up to 6 mg | Chinese adults with overweight or obesity | At 24 weeks: -6.7% with 3 mg, -10.4% with 4.5 mg, -11.3% with 6 mg, and +1.0% with placebo | Early proof that dual GLP-1/glucagon activation had meaningful weight-loss potential |
| GLORY-1, 4 mg and 6 mg | 610 Chinese adults with obesity or overweight plus at least one related comorbidity | At 48 weeks: -12.05% with 4 mg, -14.84% with 6 mg, and -0.47% with placebo under the efficacy estimand | Core Phase 3 evidence for China’s weight-management approval |
| GLORY-1 responder results | Same trial population | At 48 weeks, 37.0% with 4 mg and 50.6% with 6 mg achieved at least 15% weight loss, versus 2.1% with placebo | Shows clinically meaningful response in many, not all, participants |
| GLORY-2, 9 mg | 462 Chinese adults with obesity, including some with type 2 diabetes | At 60 weeks: -18.55% with 9 mg versus -3.02% with placebo; in participants without type 2 diabetes, mean loss reached 20.08% | Most striking obesity result, but the 9 mg dose is a higher-dose, separate regulatory question |
| Liver-fat subgroup signals | Participants with elevated liver fat in GLORY-1 and GLORY-2 subgroups | Large liver-fat reductions were reported, including -73.18% at 6 mg in GLORY-1 among those with baseline liver fat at least 5% | Promising metabolic signal, but not the same as a dedicated MASH treatment claim |
The most useful takeaway is that mazdutide has a clear weight-loss signal across several dose levels, but its strongest available evidence is still concentrated in Chinese populations and Chinese regulatory pathways.
What dose of mazdutide is being studied or approved?
Mazdutide dosing depends on country, indication, and label. Public trial evidence most often discusses once-weekly subcutaneous 4 mg and 6 mg dosing for broad weight management, while 9 mg is being evaluated for adults with moderate-to-severe obesity.
| Dose context | What was studied or used | Why it matters |
|---|---|---|
| Phase 2 obesity | 3 mg, 4.5 mg, and 6 mg weekly in one part of the trial; higher-dose work explored 9 mg and 10 mg in separate research | Dose-finding helped define efficacy and tolerability before Phase 3 |
| GLORY-1 obesity | 4 mg and 6 mg weekly versus placebo for 48 weeks | Primary evidence layer for the chronic weight-management indication in China |
| GLORY-2 obesity | 9 mg weekly versus placebo for 60 weeks | Designed for adults with higher BMI and larger weight-loss needs |
| Type 2 diabetes studies | 4 mg and 6 mg weekly in major Phase 3 diabetes programs | Diabetes outcomes are related, but they should not be treated as the same as obesity-only treatment goals |
Study doses are not self-use instructions. Approved dosing should come from the local prescribing label and a clinician who can assess medical history, medications, gallbladder risk, pancreatitis history, diabetes treatment, and tolerability.
How long does mazdutide take to work?
Mazdutide is a long-term weight-management medication, not a quick fat-loss product. The key obesity trials measured results over 24, 32, 48, and 60 weeks, with weight loss generally continuing over the trial period.
| Time point | What it tells you | Practical interpretation |
|---|---|---|
| First weeks | Appetite and gastrointestinal tolerance may change before major scale movement | Early nausea or appetite suppression is not the same as durable success |
| 24 to 32 weeks | Phase 2 and GLORY-1 primary obesity endpoints showed clinically meaningful loss | This is a more realistic checkpoint than one or two doses |
| 48 weeks | GLORY-1 reported stronger longer-term reductions at both 4 mg and 6 mg | Long-term adherence and tolerability matter |
| 60 weeks | GLORY-2 reported continued loss with 9 mg and no plateau at week 60 | Higher-dose results need dose-specific approval and label context |
A fair assessment should track more than scale weight: waist circumference, food tolerance, side effects, strength, protein intake, blood pressure, glucose markers, and whether the medication can be used safely over time.
What side effects can mazdutide cause?
The most common mazdutide side effects are gastrointestinal, similar to many GLP-1-based therapies. Nausea, diarrhea, vomiting, decreased appetite, constipation, and related symptoms are the main tolerability pattern to understand.
| Safety theme | What trials suggest | Practical interpretation |
|---|---|---|
| Gastrointestinal symptoms | Nausea, diarrhea, and vomiting were frequently reported and generally described as mild to moderate and transient | Can still affect hydration, protein intake, medications, and adherence |
| Dose and titration | Higher doses may produce stronger weight loss but can increase tolerability pressure | Dose escalation is a medical protocol, not a self-experiment |
| Discontinuation | GLORY-1 reported low adverse-event discontinuation rates; GLORY-2 reported 2.9% discontinuation due to adverse events in the 9 mg group | Low trial discontinuation does not remove the need for screening and follow-up |
| Heart rate and cardiometabolic context | GLORY-1 reported a mean heart-rate increase of 2.6 beats per minute at week 48 and no observed cardiovascular-risk safety signal in that study | Reassuring trial signal, but individual risk still matters |
Common does not mean harmless. Severe abdominal pain, persistent vomiting, dehydration, fainting, allergic symptoms, or severe low-blood-sugar symptoms should be treated as medical territory.
Mazdutide troubleshooting: what if results or side effects do not make sense?
The safest troubleshooting frame is not “adjust the dose yourself.” It is “protect safety, confirm the right medication pathway, and track objective trends under clinician supervision.”
Common mistakes
- Comparing mazdutide 9 mg data with mazdutide 4 mg or 6 mg data as if they were the same population and regulatory status.
- Comparing mazdutide with Wegovy, Ozempic, Mounjaro, or Zepbound without matching dose, indication, country, trial duration, and participant profile.
- Ignoring muscle-preserving habits during appetite suppression: protein, resistance training, hydration, sleep, and nutrient-dense meals.
Clean test protocol
| Inputs | Clinician-supervised prescription pathway, baseline weight, waist, blood pressure, medication list, diabetes status, gallbladder and pancreatitis history, liver markers when relevant, protein target, hydration plan, and resistance-training baseline. |
|---|---|
| Duration | Use the local prescribing label and clinician plan. For obesity medications, meaningful interpretation usually takes weeks to months, not a few daily weigh-ins. |
| 3 metrics | Weekly average weight trend, waist circumference trend, and side-effect burden with hydration and food-tolerance notes. |
| Stop conditions | Stop self-adjusting and seek medical guidance for severe nausea, persistent vomiting, dehydration, fainting, severe abdominal pain, allergic symptoms, chest pain, jaundice, severe low-blood-sugar symptoms, or any symptom that feels unsafe. |
How to tell it’s working
A useful response is not only a lower scale number. Better signals include steadier appetite, gradual waist reduction, manageable side effects, preserved strength where possible, better blood-pressure or glucose markers when tracked, and the ability to keep nutrition quality intact.
Red flags / seek care
Seek care for persistent vomiting, severe or persistent abdominal pain, signs of dehydration, fainting, chest pain, yellowing skin or eyes, severe allergic symptoms, confusion, severe weakness, severe low-blood-sugar symptoms, or thoughts of self-harm.
How does mazdutide work compared with semaglutide or tirzepatide?
Mazdutide is different because it targets GLP-1 and glucagon receptors. Semaglutide targets GLP-1 only, while tirzepatide targets GIP and GLP-1; those mechanism differences are meaningful, but they do not automatically prove that one drug is best for every person.
| Medication type | Main receptor targets | What to keep in mind |
|---|---|---|
| Mazdutide | GLP-1 and glucagon | Weight-loss plus liver-fat and metabolic-health rationale, with strongest data in Chinese trials |
| Semaglutide | GLP-1 | Much broader global prescribing experience depending on product and indication |
| Tirzepatide | GIP and GLP-1 | Very strong obesity data in approved markets, but not the same mechanism as mazdutide |
| Retatrutide-style multi-agonists | GIP, GLP-1, and glucagon | Investigational in many settings; mechanism excitement still needs trial and approval context |
The glucagon receptor is the key differentiator. It is being used to pursue energy expenditure and liver-fat metabolism, but higher metabolic ambition also means safety, heart rate, tolerability, and long-term outcomes deserve careful follow-up.
Is mazdutide approved and available now?
Mazdutide is approved in China for chronic weight management in eligible adults and for glycemic control in adults with type 2 diabetes. Its 9 mg dose strategy for moderate-to-severe obesity has been accepted for regulatory review in China, but that is separate from the already approved lower-dose pathway.
Outside China, mazdutide should still be treated as unavailable through routine regulated prescribing unless a local regulator has approved it. In the United States, online mazdutide products should not be treated as FDA-approved medication.
Decision rule
Approved in one country does not mean approved everywhere. The safe decision depends on your local label, your clinician, your medical history, and whether the product comes through a regulated prescription channel.
Selected Professional References
These sources support the weight-loss results, mechanism framing, side-effect interpretation, regulatory status, and safety cautions used in this guide.
New England Journal of Medicine: GLORY-1
Supports the 4 mg and 6 mg Phase 3 obesity evidence in Chinese adults with overweight or obesity.
Used for: primary weight-loss efficacy and tolerability context
Innovent GLORY-1 NEJM Publication Announcement
Supports GLORY-1 percent weight-loss values, responder rates, liver-fat reductions, and cardiometabolic endpoint notes.
Used for: detailed GLORY-1 result table
Innovent GLORY-2 9 mg Topline Results
Supports the 60-week 9 mg obesity results, 20% responder data, liver-fat subgroup, and adverse-event discontinuation rate.
Used for: higher-dose obesity evidence
Nature Communications: Phase 2 Mazdutide Trial
Supports the 24-week phase 2 weight-loss results, dose-finding context, and early safety pattern.
Used for: early efficacy and dose-response framing
Nature: Mazdutide Versus Placebo in Type 2 Diabetes
Supports the 4 mg and 6 mg diabetes evidence, weight-loss effect in type 2 diabetes, and common adverse-event pattern.
Used for: diabetes and safety context
Mazdutide: First Approval
Supports the China approval timeline and indication context for long-term weight management and later glycemic control.
Used for: approval status summary
ClinicalTrials.gov: GLORY-1
Supports study identification, population, intervention arms, and trial-design verification.
Used for: trial design context
FDA Warning Letter Referencing Online Mazdutide Products
Supports the caution that online mazdutide products marketed in the United States should not be treated as FDA-approved medication.
Used for: unapproved-product safety framing
Go Deeper (VerifiedSupps Guides)
Use these related guides to compare mazdutide with nearby GLP-1, glucagon, and next-generation obesity medication topics without mixing up mechanisms, trial stages, and approved use.
Pemvidutide for Weight Loss and Fatty Liver
Compare another GLP-1/glucagon dual agonist with a stronger liver-focused development path.
Survodutide for Weight Loss
A nearby glucagon and GLP-1 candidate with obesity and metabolic-health relevance.
Retatrutide: Weight Loss, Side Effects, and Status
Compare mazdutide with a triple-agonist research path while keeping trial stage clear.
CagriSema for Weight Loss
Understand a different combination strategy: GLP-1 plus amylin instead of GLP-1 plus glucagon.
Final Takeaway
Mazdutide is a serious GLP-1/glucagon weight-loss medication with strong China-based clinical data, clear gastrointestinal tolerability considerations, and an interesting liver-fat rationale. The calm decision is this: view it as regulated prescription medicine where approved, not as a peptide-store shortcut, and compare it against available approved options with a clinician.
FAQ
What is mazdutide?
Mazdutide is a once-weekly GLP-1/glucagon dual receptor agonist, also known as IBI362 or LY3305677. It is approved in China for specified weight-management and type 2 diabetes indications.
Is mazdutide approved for weight loss?
Yes, mazdutide is approved in China for chronic weight management in eligible adults. It is not universally approved globally, and availability depends on local regulatory status.
Is mazdutide FDA approved?
No. Mazdutide is not FDA-approved for weight loss or diabetes treatment in the United States at the time of this review.
How much weight did people lose with mazdutide?
In GLORY-1, mazdutide 6 mg showed 14.84% mean weight loss at 48 weeks under the efficacy estimand. In GLORY-2, mazdutide 9 mg showed 18.55% mean weight loss at 60 weeks, with 20.08% loss in participants without type 2 diabetes.
What are the main side effects of mazdutide?
The main side effects are gastrointestinal, including nausea, diarrhea, vomiting, decreased appetite, and constipation. Most trial-reported events were mild to moderate, but persistent symptoms should be handled medically.
What dose of mazdutide was studied?
Major trials studied once-weekly 4 mg and 6 mg doses in GLORY-1, while GLORY-2 studied 9 mg in adults with obesity. Study doses are not self-use instructions.
How long does mazdutide take to work?
Trial results were measured over months. Key endpoints appeared at 24 to 32 weeks, while stronger longer-term results were reported at 48 weeks in GLORY-1 and 60 weeks in GLORY-2.
How does mazdutide work?
Mazdutide activates GLP-1 and glucagon receptors. GLP-1 signaling supports appetite control and glucose regulation, while glucagon signaling is being used to target energy expenditure and hepatic fat metabolism.
Does mazdutide help liver fat?
Mazdutide trials reported meaningful liver-fat reductions in subgroup analyses. That is promising, but liver-fat improvement is not the same as a dedicated MASH treatment claim or a substitute for liver-disease diagnosis.
Can you buy mazdutide online?
Online mazdutide research products are not the same as regulated prescription medication. Do not treat research-vial products as safe or approved substitutes for clinician-supervised care.
VerifiedSupps Medical Disclaimer
Mazdutide is a regulated prescription medication in approved markets, not a dietary supplement. This article is for educational purposes only and does not provide medical advice, diagnosis, prescribing guidance, dosing instructions, injection instructions, or a recommendation to purchase unapproved products. Availability, indications, dose, contraindications, and monitoring requirements depend on local regulatory approval and a licensed clinician’s assessment. Obesity medications can cause meaningful side effects and may be unsafe with certain medical conditions or medications, especially diabetes medications, pregnancy considerations, gallbladder disease, pancreatitis history, kidney disease, liver disease, severe gastrointestinal symptoms, or complex cardiovascular risk. Seek urgent care for severe abdominal pain, persistent vomiting, dehydration, fainting, chest pain, jaundice, severe allergic symptoms, severe low-blood-sugar symptoms, confusion, or thoughts of self-harm.