By VerifiedSupps Editorial Team
How Retatrutide Works: GLP-1, GIP, and Glucagon Explained
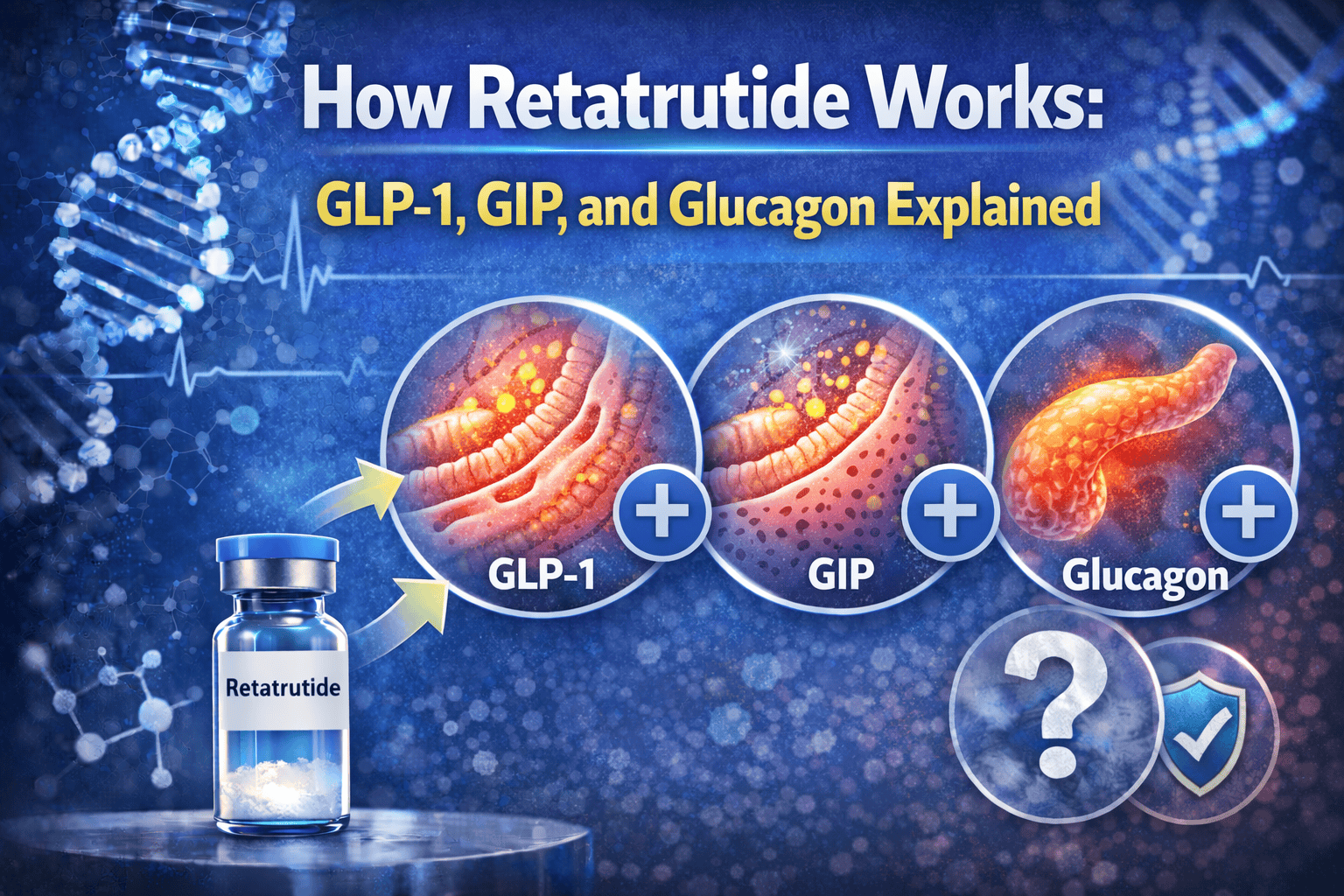
Retatrutide is getting attention because it is not just another GLP-1 drug. It is a once-weekly triple agonist that activates three receptors at the same time: GIP, GLP-1, and glucagon.
That matters because each receptor appears to contribute something different. GLP-1 helps explain the familiar appetite and gastric-emptying effects. GIP helps explain why this is not just semaglutide with extra marketing. Glucagon is the most counterintuitive piece, and probably the one most responsible for why retatrutide may behave differently from the current GLP-1 class.
This page is for the actual mechanism question: what each receptor is doing, why Lilly built the molecule this way, and how that may connect to retatrutide’s unusually strong obesity signals.
Key terms: retatrutide, triple agonist, GLP-1, GIP, glucagon, once-weekly injection, appetite, insulin secretion, energy expenditure, obesity
Quick Take
Retatrutide works by combining three hormone signals in one molecule, not by pushing one receptor harder and harder. The most useful mental model is this: GLP-1 helps explain fullness, GIP helps explain why the drug is more than a simple GLP-1 clone, and glucagon may be the piece that changes the energy-balance story most.
TL;DR decision
Retatrutide is best understood as a broader metabolic design, not just a stronger GLP-1. That broader design is exactly why the obesity results look so interesting and why the tolerability and long-term questions still matter.
Evidence standard: human trials, dose ranges, guideline-level sources when available
Who this is for: people trying to understand what retatrutide is actually doing at the receptor level and why it may behave differently from current GLP-1 drugs
Who this is not for: anyone looking for gray-market peptide guidance, trial shortcuts, or proof that mechanism alone settles clinical value
Reviewed by: VerifiedSupps Editorial Team
Last reviewed: April 17, 2026
Parent Hub
VerifiedSupps Articles
Browse the broader library if you want to compare newer metabolic therapies with calmer, more evidence-based context.
Retatrutide mechanism decoder: what the three hormones actually mean
This is the fastest way to stop hearing “triple agonist” as a buzzword and start hearing what each signal is supposed to contribute.
| If you hear… | Better translation | Why it matters | Best honest read |
|---|---|---|---|
| “It’s basically just a stronger GLP-1.” | No. GLP-1 is only one third of the design. | You miss why GIP and glucagon are there at all. | Too simplistic |
| “GIP does nothing useful here.” | Lilly frames GIP as part of insulin secretion, food intake, and lipid-metabolism control. | It helps explain why retatrutide is not just semaglutide with a different name. | Underrated in casual explanations |
| “Glucagon sounds like the opposite of a weight-loss receptor.” | Classically it raises hepatic glucose output, but in this context it is also tied to energy expenditure and lipid metabolism. | This is the part most likely to explain why retatrutide may push beyond standard GLP-1 results. | Counterintuitive but important |
| “GLP-3 is the real scientific name.” | No. It is an informal media nickname. | The cleaner term is triple agonist. | Use with caution |
Best next step (today): When you hear “retatrutide,” mentally translate it into “GLP-1 for satiety, GIP for metabolic support, glucagon for the extra energy-balance twist.”
How does retatrutide work?
Retatrutide works by activating three metabolic hormone receptors at the same time: GIP, GLP-1, and glucagon. That is the big picture. The more useful detail is that Lilly did not simply stack three strong hormone signals on top of each other. The molecule has its own balance and receptor profile.
Lilly Medical describes retatrutide as a synthetic molecule with a C20 fatty diacid moiety that helps it bind albumin and last long enough for once-weekly dosing. The same explainer says the molecule is more potent than native human GIP at the human GIP receptor, but less potent than native GLP-1 and glucagon at their own receptors.
Mechanism
- It is a true triple agonist, not a GLP-1-only drug wearing a more complex label.
- Its design supports once-weekly subcutaneous dosing because the half-life is about 6 days.
- The built-in receptor balance is one reason the molecule may behave differently from both semaglutide and tirzepatide.
What would change my recommendation: more public mechanistic human data explaining which receptor is doing how much of the real-world clinical work, rather than only relying on broad biological logic.
What does GLP-1 do in retatrutide?
GLP-1 is the most familiar part of the retatrutide story. It gives the molecule its “I already know what this kind of drug does” backbone: more fullness, slower stomach emptying, and better glucose-dependent insulin support.
This is the piece that makes retatrutide feel somewhat understandable to anyone who already knows semaglutide. Lilly Medical describes GLP-1’s role here as enhancing insulin secretion in a glucose-dependent way, modulating glucagon secretion, delaying gastric emptying, and increasing satiety signals in the brain.
If you took GLP-1 out of retatrutide, the drug would lose the most familiar appetite-and-fullness logic that made the modern obesity class work in the first place.
What does GIP do in retatrutide?
GIP is the piece that keeps retatrutide from being “just semaglutide plus hype.” Its role is less intuitive to most readers, but it matters because Lilly’s own mechanism explainer frames GIP as more than a background incretin.
According to Lilly Medical, GIP primarily enhances glucose-dependent insulin secretion, but it also decreases food intake and regulates lipid metabolism. That means it may support both glucose handling and the broader metabolic environment in which weight loss is happening.
This is one reason retatrutide should not be explained as “GLP-1 does the real work and the rest is decoration.” The molecule was built on purpose to make the GIP signal part of the main story.
What does glucagon add to retatrutide?
Glucagon is the most counterintuitive part of the entire molecule, and probably the most important part for understanding why retatrutide may not behave like a standard GLP-1 drug. Most people hear “glucagon” and think “raise glucose,” which is not wrong, but it is incomplete.
Lilly Medical notes that glucagon is well known for increasing hepatic glucose production during hypoglycemia, but also says it can increase insulin secretion, reduce food intake, modulate energy expenditure and lipid metabolism, and delay gastric emptying. In preclinical models, the extra weight-loss edge of retatrutide is often tied to glucagon receptor agonism helping preserve or raise energy expenditure during weight loss.
That is the twist. GLP-1 helps explain why people may eat less. Glucagon may help explain why the energy-balance picture looks more aggressive than a simpler GLP-1 setup. It is not the easy part of the story, but it is probably the most important if you are asking why retatrutide might outperform simpler drugs.
The right conclusion is not “glucagon is good” or “glucagon is bad.” It is that glucagon changes the design logic from appetite support alone to a broader metabolic strategy.
How much weight loss has retatrutide shown so far?
Enough to make the mechanism discussion feel clinically relevant instead of academic. The most important published obesity number is still up to 24.2% average weight loss at 48 weeks in the phase 2 trial.
Published obesity data
24.2%
Average weight loss at 48 weeks in phase 2
Phase 3 obesity topline
28.7%
Average weight loss at 68 weeks in TRIUMPH-4
Phase 3 diabetes topline
16.8%
Average weight loss at 40 weeks in first phase 3 T2D study
That matters because the mechanism explanation is not being attached to a weak or trivial outcome signal. It is being attached to one of the strongest obesity drug signals currently in development. The caution is simply that the strongest newer numbers are still partly living in topline-release territory rather than in fully published final detail.
Is retatrutide approved yet?
No. Retatrutide is still investigational, not approved, and legally available only to participants in Lilly-sponsored clinical trials. That part of the story should stay very simple.
The more nuanced part is that “not approved” does not mean “barely studied.” Lilly says retatrutide has already successfully completed two phase 3 trials and is still being studied across obesity, type 2 diabetes, knee osteoarthritis pain, sleep apnea, chronic low back pain, cardiovascular and renal outcomes, and metabolic liver disease.
So the current status is not early hype. It is late-stage development. But late-stage development is still not the same thing as a finished public-use therapy.
What should you do if retatrutide sounds like “just a stronger GLP-1”?
The first step is to stop flattening the molecule. Retatrutide is not best understood as “GLP-1, but more.” It is better understood as an attempt to build appetite, insulin, lipid, and energy-balance effects into one coordinated design.
Common mistakes
- Assuming GLP-1 explains the whole drug and treating GIP and glucagon like background noise.
- Treating “triple agonist” as proof of superiority before the full clinical evidence stack is finished.
- Using the informal “GLP-3” label as if it were a scientific category instead of media shorthand.
- Thinking mechanism alone settles the safety, tolerability, and approval questions.
Clean test protocol
| Inputs | A clear understanding of what GLP-1, GIP, and glucagon each contribute, plus a clear distinction between mechanistic promise and current clinical status |
|---|---|
| Duration | Reassess when more full phase 3 publications arrive and when direct comparisons versus current obesity leaders are more complete |
| 3 metrics | Whether the receptor explanation now makes sense, whether you can separate mechanism from marketing, and whether you understand why approval status still matters |
| Stop conditions | Any impulse to treat an investigational mechanism story like a green light for buying fake or unverified “retatrutide” online |
How to tell it’s working
Right now, the more useful test is whether your model of the drug is more accurate. A good mechanism explainer should leave you less impressed by buzzwords and more able to understand why retatrutide looks distinct without acting like the case is already closed.
Red flags / seek care
If you are using any unverified product claimed to be retatrutide and develop persistent vomiting, severe dehydration, chest pain, trouble breathing, severe palpitations, or a strong allergic-type reaction, stop and seek medical care immediately. Fake-source risk is part of the safety story here.
Selected Professional References
These are the most useful sources for understanding what each receptor is supposed to contribute, why the mechanism matters, and how it connects to the current obesity evidence.
What to Know About Retatrutide
The cleanest official source for what retatrutide is, why “GLP-3” is not a scientific label, and where the drug currently stands.
Used for: current status, trial-only access, and the “triple agonist” framing
Lilly Medical: Mechanism of Action of Retatrutide
The most useful source for what GLP-1, GIP, and glucagon are each supposed to contribute.
Used for: receptor roles, half-life, and receptor-potency profile
Triple–Hormone-Receptor Agonist Retatrutide for Obesity
Still the most important published source connecting the mechanism story to real obesity outcomes.
Used for: strongest published obesity efficacy context
Retatrutide in Type 2 Diabetes
Useful for the early clinical context behind why the mechanism looked worth pushing forward.
Used for: early glucose and weight signal in humans
Triple Agonism Based Therapies for Obesity
A good review-level explanation of why the glucagon piece is such a big deal in the triple-agonist concept.
Used for: why triple agonism may push beyond simpler incretin designs
TRANSCEND-T2D-1 Results
Helpful for showing the mechanism discussion now sits inside a more mature phase 3 story, not just phase 2 curiosity.
Used for: current phase 3 diabetes efficacy context
TRIUMPH-4 Results
A useful reminder that the mechanism conversation now matters because the obesity numbers are becoming unusually strong.
Used for: strongest current public obesity number
Go Deeper (VerifiedSupps Guides)
These are the best next reads if you want the retatrutide cluster to make more sense after the mechanism is clear.
Retatrutide for Weight Loss
Best next read if you want the obesity-results angle after understanding the mechanism.
Retatrutide: Results, Side Effects, and Current Status
Helpful if you want the cleaner status snapshot after this receptor-level explainer.
Retatrutide vs Tirzepatide
Useful if you want to see how the triple-agonist idea compares with the current dual-agonist leader.
Retatrutide vs Semaglutide
Helpful if you want to compare the triple-agonist model with the current single-receptor obesity benchmark.
Final Takeaway
Retatrutide works by doing something more ambitious than a standard GLP-1 drug. GLP-1 helps explain satiety and gastric-emptying effects, GIP helps explain why the drug is not just a relabeled GLP-1, and glucagon may be the piece that changes the energy-balance story most. That does not automatically make retatrutide “better” in every practical sense right now. But it does explain why the mechanism and the obesity results are getting so much attention at the same time.
FAQ
How does retatrutide work?
Retatrutide works by activating three receptors at once: GIP, GLP-1, and glucagon. The goal is to combine appetite effects, insulin support, and broader energy-balance effects in one molecule.
Is retatrutide just a stronger GLP-1 drug?
No. GLP-1 is only one part of the mechanism. GIP and glucagon are there for a reason, and they are part of what makes the drug distinct.
What does GLP-1 do in retatrutide?
GLP-1 supports glucose-dependent insulin secretion, slows gastric emptying, modulates glucagon secretion, and increases satiety signals in the brain.
What does GIP do in retatrutide?
GIP primarily enhances glucose-dependent insulin secretion and is also linked to food-intake reduction and lipid-metabolism regulation in Lilly’s mechanism explanation.
What does glucagon add to retatrutide?
Glucagon is the most counterintuitive part of the design. In addition to its classic glucose-raising role during hypoglycemia, it is also linked to reduced food intake, energy expenditure, and lipid metabolism, which may help explain why retatrutide can look stronger than simpler GLP-1 therapies.
Why might retatrutide cause more weight loss than GLP-1 alone?
Because the triple-agonist design may combine satiety and gastric-emptying effects with broader metabolic and energy-expenditure effects, especially through the glucagon component.
How much weight loss has retatrutide shown so far?
The best published obesity result so far is up to 24.2% average weight loss at 48 weeks in phase 2, with newer public phase 3 toplines showing even larger numbers.
Is retatrutide approved yet?
No. Retatrutide is still investigational and is legally available only to participants in Lilly-sponsored clinical trials.
Is GLP-3 the real scientific name for retatrutide?
No. Lilly says GLP-3 is a scientifically inaccurate informal media nickname. Triple agonist is the more accurate term.
What is the safest way to think about retatrutide today?
Treat it as a broad metabolic design with strong obesity promise, but still as an investigational medicine whose long-term story is not fully finished.
VerifiedSupps Medical Disclaimer
This content is for educational purposes only and is not medical advice. Retatrutide is an investigational medicine, not an approved public-use therapy. Mechanism should not be confused with proof of long-term benefit, safety, or access. Do not use unverified products sold as retatrutide as a substitute for medical care, approved obesity treatment, or legitimate clinical-trial participation. Seek urgent medical care for persistent vomiting, severe dehydration, chest pain, trouble breathing, severe palpitations, or a strong allergic-type reaction after using any unverified injectable product.