CT-388 vs Tirzepatide: How Roche’s GLP-1/GIP Drug Compares
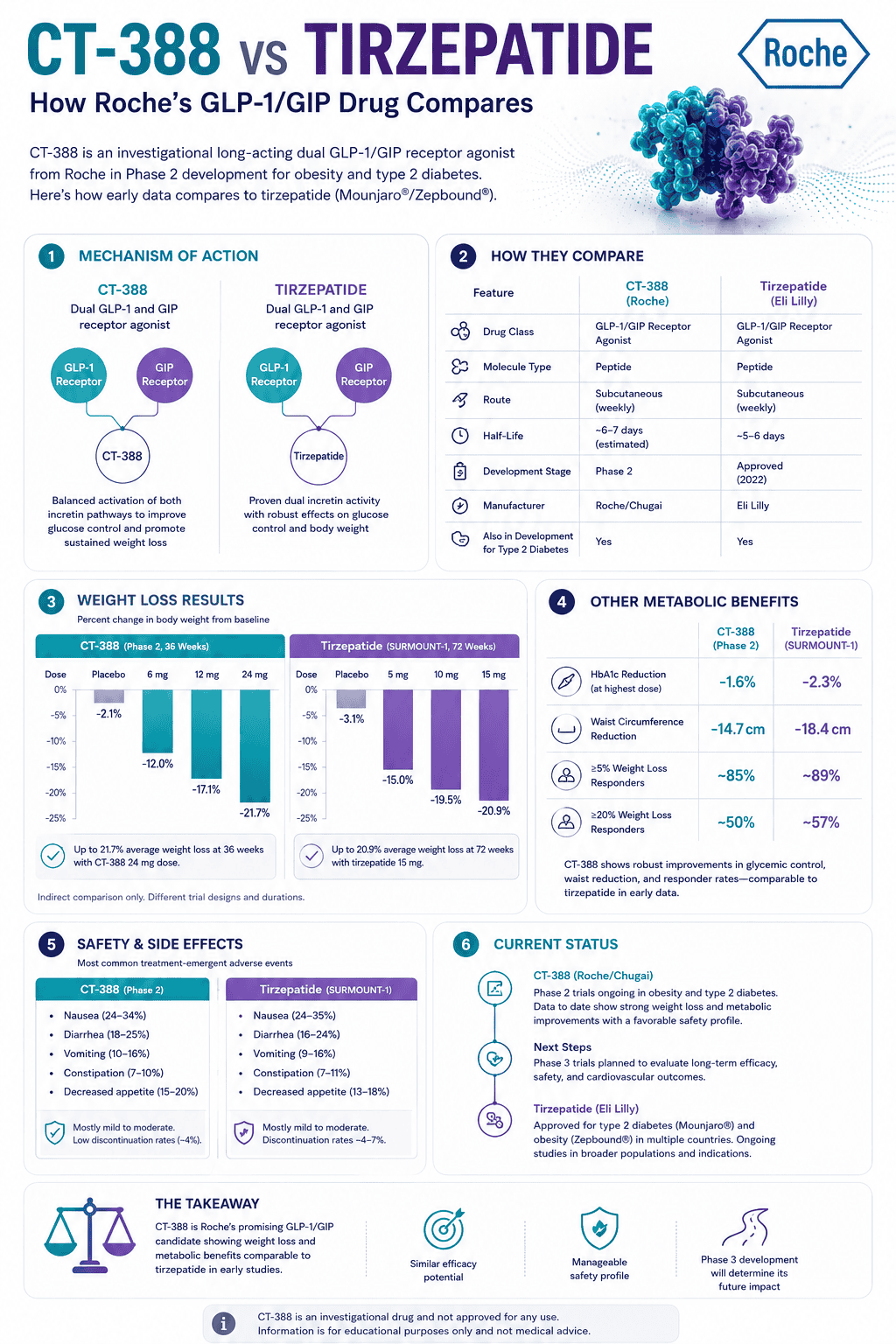
CT-388 and tirzepatide are both once-weekly injectable GLP-1/GIP receptor agonists, but they are not at the same evidence stage. Tirzepatide is FDA-approved for chronic weight management under the Zepbound brand, while CT-388 is still investigational.
CT-388 has shown strong Phase 2 weight-loss results, including 22.5% placebo-adjusted weight loss at 48 weeks at the highest tested dose. That does not prove it is better than tirzepatide, because the drugs have not been compared head-to-head and the trials used different designs.
This comparison focuses on publicly available CT-388 Phase 1/2 and pipeline data, tirzepatide Phase 3 obesity data, and current U.S. prescribing information through May 13, 2026.
Who this is for: Readers comparing Roche’s CT-388 with tirzepatide for weight-loss efficacy, side effects, dosing, and approval status.
Who this is not for: Anyone trying to buy, compound, or use CT-388 outside a regulated clinical trial.
Reviewed by VerifiedSupps Editorial Team • Last reviewed: May 13, 2026
The most important answer is simple: tirzepatide is the more established option today, while CT-388 is a promising competitor still in development. If someone is making a real treatment decision now, tirzepatide has the advantage because it has completed large Phase 3 trials and has an FDA-approved prescribing label.
CT-388 is worth watching because its reported Phase 2 results are strong, especially the 48-week placebo-adjusted weight-loss figure. But CT-388’s public dataset is still smaller, less mature, and not yet supported by completed Phase 3 obesity outcomes.
The fairest interpretation is not “CT-388 beats tirzepatide.” It is: CT-388 may become a serious GLP-1/GIP competitor if Phase 3 results confirm the Phase 2 signal, tolerability holds up, and regulators approve it.
For more background on CT-388 alone, see our companion article on CT-388 for weight loss. For a broader incretin-drug comparison, see retatrutide vs tirzepatide.
Key Takeaways
- CT-388 and tirzepatide both target GLP-1 and GIP receptors, but they are different molecules with different evidence packages.
- Tirzepatide is FDA-approved for chronic weight management; CT-388 is investigational and not commercially available.
- CT-388 reported 22.5% placebo-adjusted weight loss at 48 weeks in Phase 2, but this is not directly comparable to tirzepatide’s Phase 3 results.
- In SURMOUNT-1, tirzepatide 15 mg produced 20.9% mean weight loss from baseline at 72 weeks, versus 3.1% with placebo.
- Both drugs are associated mainly with gastrointestinal side effects, but tirzepatide has more complete safety labeling and longer clinical experience.
- CT-388’s next major test is Phase 3 confirmation, including durability, tolerability, and performance in people with and without type 2 diabetes.
Is CT-388 better than tirzepatide for weight loss?
No direct evidence shows that CT-388 is better than tirzepatide. CT-388 has promising Phase 2 results, but tirzepatide has stronger clinical evidence because it has completed large Phase 3 trials and is already approved.
The comparison is tempting because both drugs are dual GLP-1/GIP receptor agonists. But cross-trial comparisons can mislead, especially when one result is reported as placebo-adjusted weight loss and another is reported as mean weight loss from baseline.
Mechanism
- Both drugs activate GLP-1 and GIP pathways involved in appetite, satiety, glucose regulation, and energy intake.
- Tirzepatide is an approved dual GLP-1/GIP receptor agonist with established dosing and labeling.
- CT-388 is described as a signal-biased dual GLP-1/GIP agonist designed to limit beta-arrestin recruitment and receptor internalization.
- A mechanistic difference does not prove better real-world outcomes. Phase 3 efficacy, safety, and adherence matter more than theory.
For context on how incretin mechanisms can differ across drugs, our guide to how retatrutide works explains a related triple-agonist approach. CT-388 and tirzepatide are both dual agonists, not triple agonists.
How do CT-388 and tirzepatide results compare?
CT-388’s headline result is 22.5% placebo-adjusted weight loss at 48 weeks in Phase 2. Tirzepatide’s pivotal SURMOUNT-1 result was up to 20.9% mean weight loss from baseline at 72 weeks, compared with 3.1% for placebo.
These numbers should not be ranked as if they came from the same study. CT-388’s result is topline Phase 2 data at 48 weeks; tirzepatide’s result is peer-reviewed Phase 3 data at 72 weeks.
| Category | CT-388 | Tirzepatide |
|---|---|---|
| Developer | Roche / Genentech | Eli Lilly |
| Drug type | Dual GLP-1/GIP receptor agonist | Dual GIP/GLP-1 receptor agonist |
| Current status | Investigational; Phase 3 initiated | FDA-approved for chronic weight management as Zepbound |
| Key obesity study | CT388-103 Phase 2, 48 weeks, adults without type 2 diabetes | SURMOUNT-1 Phase 3, 72 weeks, adults without type 2 diabetes |
| Headline weight-loss result | 22.5% placebo-adjusted weight loss at 48 weeks at 24 mg | 20.9% mean weight loss from baseline at 72 weeks with 15 mg; placebo was 3.1% |
| ≥20% weight-loss responders | 47.8% at 48 weeks in the 24 mg group | 57% at 72 weeks in the 15 mg group; 50% in the 10 mg group |
| Main caution | Full Phase 3 data not yet available | GI side effects, contraindications, and long-term treatment planning |
A careful reader should focus less on which headline number is bigger and more on the maturity of the evidence. Tirzepatide has already shown efficacy across large trials and approved use. CT-388 still needs Phase 3 confirmation.
For another example of why cross-trial comparisons require caution, see our breakdown of retatrutide vs semaglutide for weight loss.
How do CT-388 and tirzepatide doses compare?
Both drugs are studied or used as once-weekly subcutaneous injections, but their milligram doses are not directly comparable. CT-388 tested up to 24 mg in Phase 2, while tirzepatide’s approved weight-management maintenance doses are 5 mg, 10 mg, or 15 mg once weekly.
A higher milligram number does not automatically mean a stronger drug. Dose depends on molecule design, exposure, receptor activity, tolerability, and how the drug is formulated.
- CT-388: Investigational once-weekly injection; Phase 2 tested low, middle, and high doses with 24 mg as the highest tested dose.
- Tirzepatide: Approved once-weekly injection; starts at 2.5 mg for 4 weeks, then increases based on response and tolerability.
- Tirzepatide maintenance: 5 mg, 10 mg, or 15 mg once weekly for weight reduction and long-term maintenance.
- Important difference: Tirzepatide has an approved dose-escalation schedule. CT-388 does not have a consumer dose or prescribing label.
This is why trial dosing should never be copied from press releases or registry entries. For broader safety context around unapproved peptide products, read our guide on whether peptides are safe.
How do CT-388 and tirzepatide side effects compare?
Both drugs appear to share the main tolerability pattern of incretin-based medicines: gastrointestinal side effects are the most common. Tirzepatide has more complete safety information because it has an FDA label and larger clinical database.
In CT-388’s Phase 2 obesity trial, Roche reported that most gastrointestinal adverse events were mild to moderate and that adverse-event discontinuation was 5.9% across CT-388 arms versus 1.3% with placebo. In SURMOUNT-1, adverse events caused treatment discontinuation in 4.3%, 7.1%, and 6.2% of tirzepatide 5 mg, 10 mg, and 15 mg participants, compared with 2.6% with placebo.
| Safety question | CT-388 | Tirzepatide |
|---|---|---|
| Most likely side-effect category | Gastrointestinal effects reported as most common in public data | Nausea, diarrhea, vomiting, constipation, abdominal pain, indigestion, reflux, and related effects |
| Discontinuation data | 5.9% due to adverse events across CT-388 arms in Phase 2 | 4.3% to 7.1% due to adverse events across SURMOUNT-1 dose groups |
| Known label warnings | No approved label yet | Boxed warning for thyroid C-cell tumors risk; contraindications for personal or family history of MTC or MEN 2 |
| Unanswered questions | Long-term safety, rare events, real-world tolerability, and Phase 3 durability | Long-term adherence, access, weight regain after stopping, and individual tolerability |
Tirzepatide labeling includes warnings and precautions for severe gastrointestinal problems, dehydration-related kidney problems, gallbladder problems, pancreatitis, serious allergic reactions, hypoglycemia when used with insulin or sulfonylureas, vision changes in people with type 2 diabetes, and aspiration risk during anesthesia or deep sedation.
Large weight loss can also include lean mass loss if protein intake, resistance training, and medical monitoring are neglected. For related context, see our articles on muscle loss with incretin-based weight loss and how much protein you actually need.
Which has stronger evidence and approval status?
Tirzepatide has stronger evidence today. It has completed pivotal Phase 3 obesity trials and is FDA-approved for chronic weight management, while CT-388 is still being tested in Phase 3.
This does not mean CT-388 is unimportant. It means the level of certainty is different.
- Tirzepatide: Approved, prescribed, labeled, and supported by large Phase 3 data.
- CT-388: Investigational, with strong Phase 2 topline data and Phase 3 ENITH studies initiated.
- Evidence gap: CT-388 needs completed Phase 3 trials, full safety tables, longer follow-up, and regulatory review.
- Head-to-head gap: There is no completed direct CT-388 vs tirzepatide obesity trial proving superiority.
Readers tracking the broader pipeline may also want to compare retatrutide trial results, MariTide for weight loss, and CagriSema for weight loss. These are different development programs, so the same caution about cross-trial comparisons applies.
Who is tirzepatide better for, and who is CT-388 for?
Tirzepatide may be considered by eligible adults working with a licensed clinician. CT-388 is only appropriate in a regulated clinical trial because it is not approved and has no final prescribing label.
The practical distinction is clear: tirzepatide is a medical treatment option; CT-388 is a clinical research candidate.
- Tirzepatide may be relevant for: adults with obesity, or adults with overweight and at least one weight-related condition, when prescribed with diet and activity support.
- CT-388 may be relevant for: people who meet eligibility criteria for a registered clinical trial and understand the investigational nature of the drug.
- Neither is a casual weight-loss shortcut: both require medical screening, side-effect monitoring, and long-term planning.
- Anyone considering gray-market CT-388: should avoid it. Online availability does not establish identity, purity, sterility, dosing accuracy, or safety.
People who are pregnant, trying to become pregnant, breastfeeding, have a history of pancreatitis or gallbladder disease, use insulin or sulfonylureas, have severe gastrointestinal motility disorders, or have active eating-disorder risk should discuss these issues carefully with a clinician before using any incretin-based obesity medication.
What should we watch next for CT-388 vs tirzepatide?
The most important next step is Phase 3 data for CT-388. If the Phase 3 ENITH program confirms strong weight loss with acceptable tolerability, CT-388 could become a serious competitor to approved GLP-1/GIP drugs.
Until then, the key questions are still open:
- Does CT-388 maintain or improve on its Phase 2 weight-loss signal over 72 weeks?
- How does CT-388 perform in people with type 2 diabetes?
- Do gastrointestinal side effects remain manageable at scale?
- Are rare safety issues detectable in larger studies?
- How much lean mass is lost, and how does that compare with other drugs?
- Will CT-388 be used alone, or as part of future combination strategies with candidates such as petrelintide?
For more pipeline context, see our guides to petrelintide for weight loss, amycretin for weight loss, and pemvidutide for weight loss and fatty liver.
References
- Genentech. Positive Phase II Results for CT-388 in People Living With Obesity. January 26, 2026.
This source supports CT-388’s 48-week Phase 2 topline results, including 22.5% placebo-adjusted weight loss, responder rates, BMI data, adverse-event discontinuation, and the statement that full results were planned for future presentation.
- Roche. Positive Phase II Results for Dual GLP-1/GIP Receptor Agonist CT-388. January 27, 2026.
This Roche release confirms CT-388’s investigational status, Phase 2 study design, highest tested 24 mg dose, dose-response language, and planned Phase 3 development.
- Genentech Medical Information. CT-388 Pipeline and Clinical Trials.
This source supports the description of CT-388 as an investigational once-weekly dual GLP-1/GIP receptor agonist and the proposed signal-biased mechanism with minimal to no beta-arrestin recruitment.
- Roche ForPatients. CT388-103 Study Details.
This trial listing supports the Phase 2 CT388-103 design, eligibility criteria, obesity and overweight population, and 48-week study structure.
- Roche Group Development Pipeline. Status as of April 23, 2026.
This pipeline document supports CT-388’s Phase 3 ENITH-1 and ENITH-2 status, including planned study populations and week-72 body-weight endpoints.
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide Once Weekly for the Treatment of Obesity. New England Journal of Medicine. 2022.
This pivotal SURMOUNT-1 publication supports tirzepatide’s 72-week weight-loss results, responder rates, and adverse-event discontinuation rates in adults with obesity or overweight without diabetes.
- U.S. Food and Drug Administration. FDA Approves New Medication for Chronic Weight Management. November 8, 2023.
This FDA announcement supports tirzepatide’s approval as Zepbound for chronic weight management in adults with obesity or overweight with at least one weight-related condition.
- DailyMed. Zepbound Prescribing Information.
This label source supports tirzepatide’s approved dosing, contraindications, common side effects, and warnings relevant to clinical use.
- Chakravarthy MV, et al. Effects of CT-388, a once-weekly signaling-biased dual GLP-1/GIP receptor agonist, on weight loss and glycemic control. Molecular Metabolism. 2025.
This peer-reviewed source supports the mechanistic framing of CT-388 as a signal-biased dual GLP-1/GIP agonist and provides early preclinical and clinical context.
FAQ
Is CT-388 the same as tirzepatide?
No. CT-388 and tirzepatide both target GLP-1 and GIP receptors, but they are different molecules from different companies. Tirzepatide is approved; CT-388 is investigational.
Is CT-388 better than Zepbound?
That is not known. CT-388 has strong Phase 2 topline results, but Zepbound has completed Phase 3 trials and has FDA approval. A direct head-to-head trial would be needed for a fair superiority claim.
Is CT-388 available to buy?
No. CT-388 is not approved and is not commercially available. Products sold online as CT-388 should be considered unverified and unsafe.
Why does CT-388’s weight-loss number look higher?
CT-388’s 22.5% figure is placebo-adjusted and comes from a Phase 2 topline report at 48 weeks. Tirzepatide’s commonly cited 20.9% figure is mean weight loss from baseline in a Phase 3 trial at 72 weeks. Those are not identical comparisons.
Does CT-388 have fewer side effects than tirzepatide?
There is not enough evidence to say that. CT-388’s early tolerability data are encouraging, but tirzepatide has a much larger safety dataset and an approved label.
Could CT-388 replace tirzepatide?
It is too early to know. CT-388 could become a competitor if Phase 3 results are strong and regulators approve it, but replacement claims are premature.
Medical Disclaimer
This article is for educational purposes only and does not provide medical advice, diagnosis, or treatment. Tirzepatide is a prescription medication that should only be used under the care of a licensed clinician. CT-388 is an investigational drug candidate and should not be purchased, compounded, prescribed, or used outside a regulated clinical trial unless it receives regulatory approval and is prescribed by a qualified clinician. People considering obesity treatment should speak with a healthcare professional about approved options, contraindications, side effects, glucose monitoring, nutrition, muscle preservation, and long-term weight-management planning.