By VerifiedSupps Editorial Team
Retatrutide: How It Works, Weight Loss Results, and What We Actually Know So Far
Retatrutide looks like one of the most powerful obesity drugs in development so far. The strongest published obesity dataset still comes from its phase 2 trial, where the highest dose reached 24.2% average weight loss at 48 weeks, and the most dramatic public phase 3 readout so far came from a knee-osteoarthritis obesity trial, where the 12 mg dose reached 28.7% at 68 weeks.
That sounds impressive because it is. But the clean reading is still more careful than the hype. Retatrutide is not approved yet, some of the biggest newer results are still topline rather than fully peer-reviewed publications, and there are still important unanswered questions around long-term safety, head-to-head performance, and who benefits most.
This page is for the broad retatrutide decision: how the drug works, what the best weight-loss results actually are, what has and has not been published, and what still should make you cautious right now.
Key terms: retatrutide, triple agonist, GIP, GLP-1, glucagon, obesity medicine, weight loss results, phase 2, phase 3
Quick Take
Retatrutide is probably the most closely watched next-generation obesity drug in development because the weight-loss numbers so far are unusually strong. But the smartest way to read it is as a very promising investigational medicine, not as a settled winner with every major question already answered.
TL;DR decision
Retatrutide looks real, not fake. The weight-loss signal is strong. But it is still investigational, still not approved, and still not something you should treat like a finished, fully settled therapy.
Evidence standard: human trials, dose ranges, guideline-level sources when available
Who this is for: people tracking newer obesity drugs and trying to understand whether retatrutide is truly different, how much weight loss it has shown, and how much caution still makes sense
Who this is not for: anyone looking for gray-market sourcing advice, fake trial shortcuts, or reassurance that anything sold online as retatrutide is legitimate
Reviewed by: VerifiedSupps Editorial Team
Last reviewed: April 16, 2026
Parent Hub
VerifiedSupps Articles
Browse the broader evidence-based library if you want a calmer, more grounded view of where newer therapies fit alongside older, more established nutrition and supplement decisions.
Retatrutide results so far: what has actually been shown?
This is the fastest way to keep the excitement attached to the right level of evidence.
| If you are asking… | Best current answer | Best data so far | Confidence |
|---|---|---|---|
| How much weight loss has retatrutide shown in obesity without diabetes? | Very strong published phase 2 signal | Up to 24.2% average weight loss at 48 weeks | Moderate to high |
| What is the biggest phase 3 weight-loss result public so far? | Very strong topline result | Up to 28.7% average weight loss at 68 weeks in obesity or overweight plus knee osteoarthritis | Moderate, because full peer-reviewed publication is still pending |
| How much weight loss has it shown in type 2 diabetes? | Strong, but smaller than obesity-only settings | Up to 16.8% average weight loss at 40 weeks in first phase 3 diabetes trial | Moderate, because full peer-reviewed publication is still pending |
| Can you get it right now like an approved drug? | No | Retatrutide is still investigational and trial-only | High |
Best next step (today): Treat the phase 2 obesity trial as the strongest published dataset, and treat newer phase 3 weight-loss headlines as promising but still not the same thing as a fully published final evidence package.
Does retatrutide actually work for weight loss?
Yes, the current evidence strongly suggests that it does. The real question is not whether retatrutide works at all. The more useful question is how much confidence we should have in the size of the effect, in which patient groups, and with what long-term tradeoffs.
Right now, the strongest published obesity dataset is its 48-week phase 2 trial in adults with obesity or overweight without diabetes. That trial showed unusually large dose-dependent weight loss, especially at 8 mg and 12 mg. Newer phase 3 public results make the story even more impressive, but those data are still partly living in topline-release territory rather than fully published peer-reviewed detail.
Mechanism
- Retatrutide activates GIP, GLP-1, and glucagon receptors in one molecule.
- The triple-agonist design appears to help by reducing food intake and, at least in preclinical work, by increasing energy expenditure.
- That makes the weight-loss story biologically plausible, but the exact contribution of each receptor in real patients is still being worked out.
What would change my recommendation: broad peer-reviewed phase 3 obesity publications, longer-term maintenance data, and the head-to-head readout versus tirzepatide.
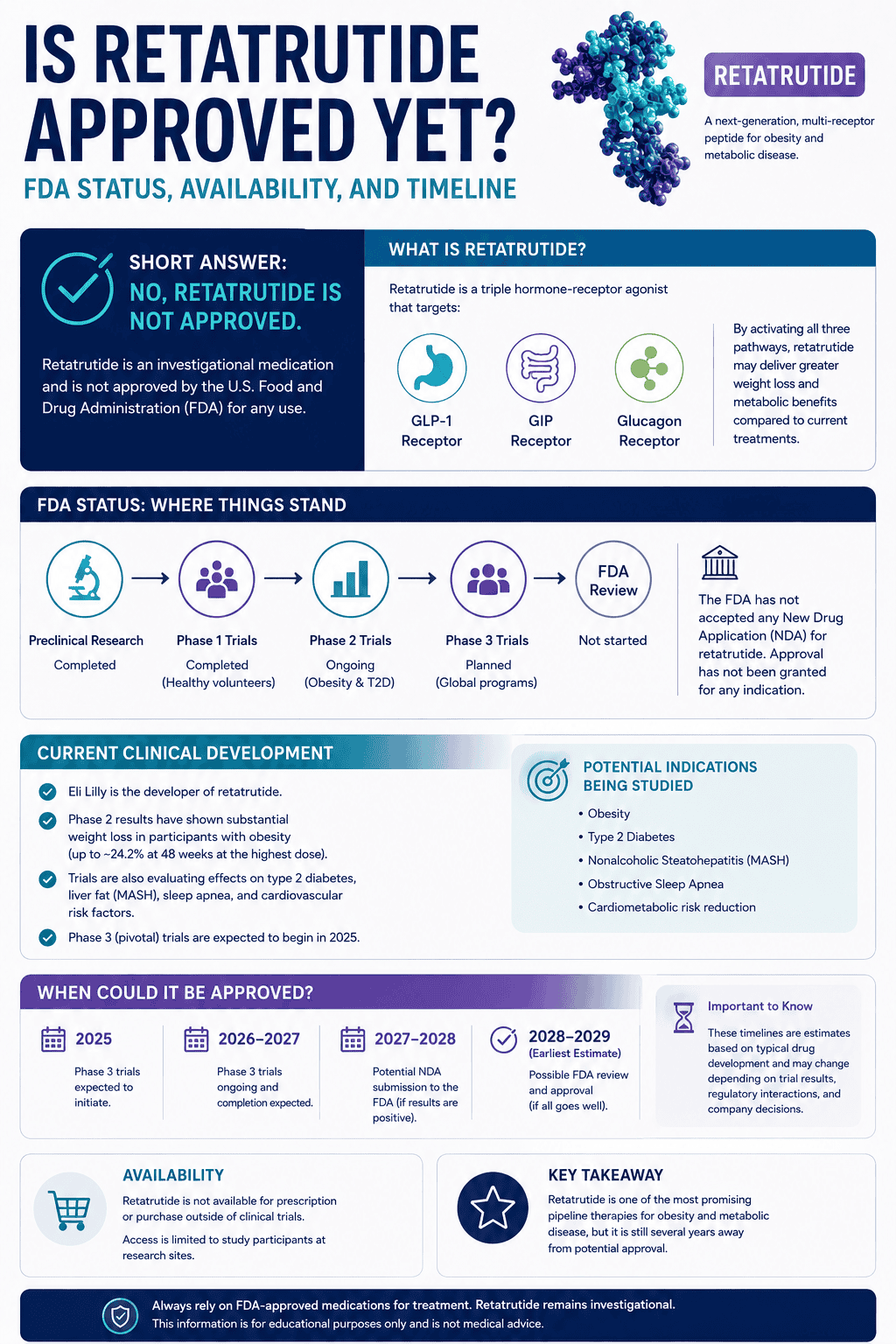
How does retatrutide work in the body?
Retatrutide is not just another GLP-1 drug. It is a triple agonist that hits GIP, GLP-1, and glucagon receptors. That is the whole reason people pay attention to it.
The practical idea is that GLP-1 and GIP effects help with appetite, food intake, and glucose control, while glucagon receptor activity may add a different metabolic layer that helps push weight loss further. In preclinical work, the glucagon component has been linked to higher energy expenditure, which is one reason retatrutide looks more aggressive than simpler incretin-based setups.
That does not mean the drug is “better” in every way yet. It means the mechanism is broader, and the early clinical data suggest that broader mechanism may translate into more weight loss. The exact balance of benefit versus tolerability is still being defined in phase 3.
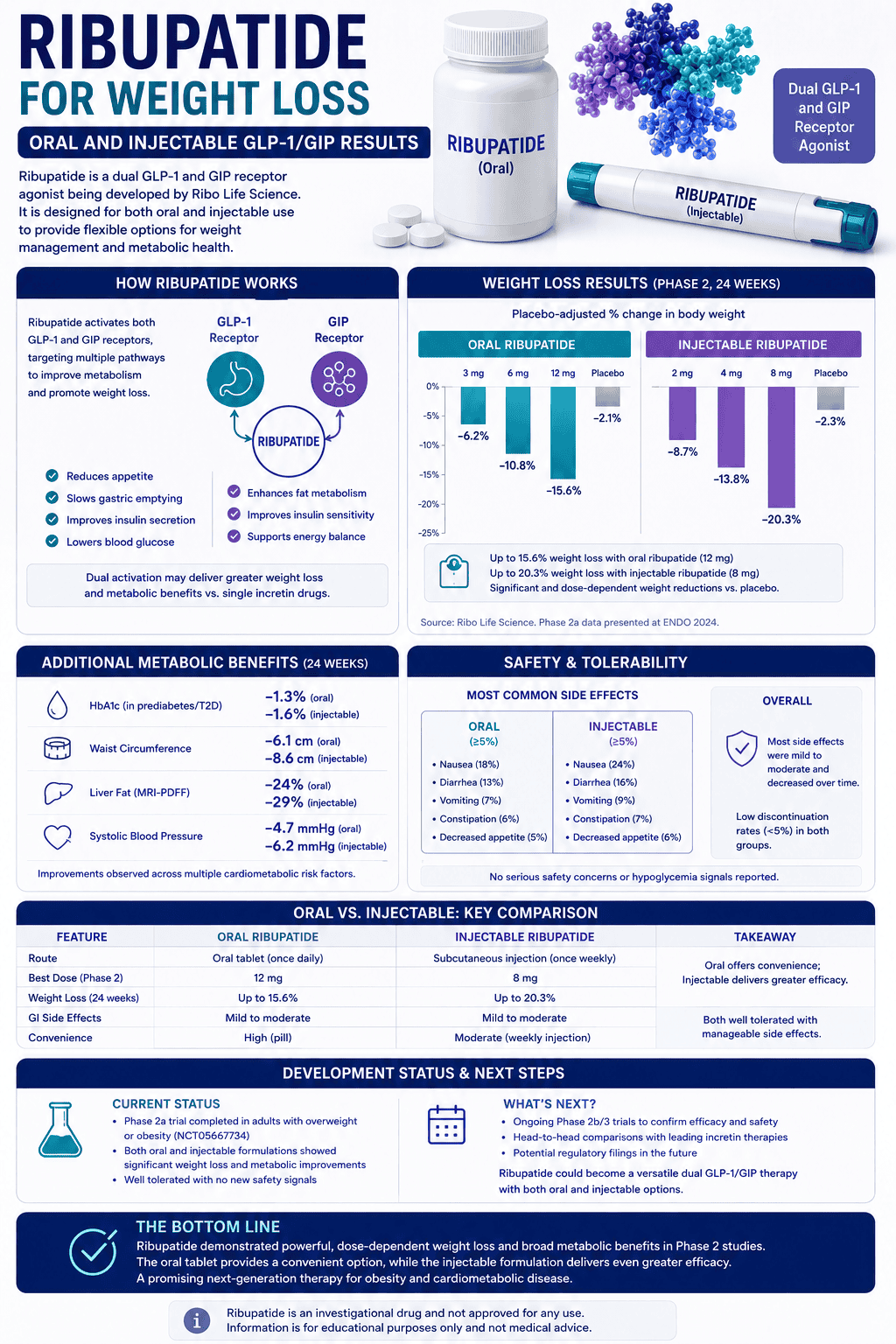
What weight loss results has retatrutide shown so far?
The cleanest way to read the data is to separate published phase 2 obesity results from newer phase 3 topline updates. That keeps you from mixing different populations, different study lengths, and different evidence tiers into one overconfident headline.
Published phase 2 obesity trial
In 338 adults with obesity or overweight without diabetes, the highest dose reached 17.5% average weight loss at 24 weeks and 24.2% at 48 weeks. That remains the best fully published obesity dataset so far.
Most important detail: the weight-loss curve still looked like it had room to run at the end of the study.
Phase 3 obesity + knee osteoarthritis topline
In TRIUMPH-4, the 12 mg dose reached 28.7% average weight loss at 68 weeks, with major improvement in knee-pain scores too.
Most important caution: these are very promising topline results, but full peer-reviewed publication is still pending.
Phase 3 type 2 diabetes topline
In TRANSCEND-T2D-1, the 12 mg dose reached 16.8% average weight loss at 40 weeks along with up to 2.0% A1C reduction.
Most important context: weight-loss results are usually smaller in type 2 diabetes than in obesity-only populations, so raw percentages are not a fair apples-to-apples comparison.
What dose of retatrutide has been studied so far?
Retatrutide is being studied as a once-weekly subcutaneous injection, but there is no approved public-use dose because the drug is still investigational. That sounds obvious, but it matters because people often speak about these regimens as if they are already standard clinical practice.
In phase 2 obesity, Lilly studied multiple regimens including 1 mg, 4 mg, 8 mg, and 12 mg, with different starting-dose strategies. In phase 3, Lilly has generally used a lower-dose start and a slow stepwise escalation every four weeks to target doses like 4 mg, 9 mg, or 12 mg depending on the trial.
The most important practical point is not “what is the best dose?” It is that the slow escalation appears to be part of how Lilly is trying to make the drug more tolerable. That alone tells you something about the side-effect burden at the higher end.
What side effects and safety concerns matter most?
The main signal so far looks like what you would expect from a very potent incretin-style drug: gastrointestinal side effects are the biggest practical issue, especially during dose escalation. The difference is that retatrutide may push that tolerability question a bit harder because its efficacy also appears unusually strong.
What shows up most often
Nausea, diarrhea, vomiting, and constipation are the main recurring adverse events. In phase 2, these were dose-related, mostly mild to moderate, and more common during escalation.
That is one reason the titration strategy matters so much.
The less talked-about signals
Phase 2 also showed a dose-dependent increase in heart rate that peaked around 24 weeks and then declined. Skin-sensation issues such as hyperesthesia or dysesthesia also appeared in a minority of participants.
These are not the headline side effects, but they are part of the real picture.
Why phase 3 still matters
The strongest weight-loss numbers are exciting, but large and longer phase 3 programs are exactly how the real tolerability and long-term risk profile gets clarified.
For now, the fairest summary is promising efficacy with a still-evolving safety picture.
Is retatrutide approved and can you get it now?
No. Retatrutide is still investigational. As of Lilly’s March 2026 FAQ update, it is not FDA-approved and is legally available only to participants in Lilly-sponsored clinical trials.
That means a clean line needs to be drawn here: current public excitement is not the same thing as current legitimate availability. Lilly explicitly warns people not to take anything claiming to be retatrutide outside its clinical trials.
So if you are reading about retatrutide as if it were simply “the next obesity shot you can get,” that is not the real status yet. The real status is late-stage development, not normal public prescribing.
What still is not known about retatrutide?
Quite a bit, and this is where the calm reading matters most. Retatrutide looks unusually strong, but the next layer of questions is exactly the layer that decides whether a drug becomes merely exciting or truly dominant.
- How it compares directly with tirzepatide: a head-to-head obesity study is ongoing, but that answer is not here yet.
- How durable the weight loss is: maintenance studies are still part of the ongoing program.
- What the long-term cardiovascular and renal picture looks like: outcome questions are still being studied.
- How well the strongest topline obesity findings generalize: the biggest public phase 3 result so far came from a specific obesity-plus-knee-osteoarthritis population.
- How tolerable the highest-efficacy regimens are in the real world: slow escalation helps, but that does not erase the side-effect burden question.
What should you do if you are thinking about retatrutide now?
The smartest move right now is not to chase it. It is to read it accurately. Retatrutide is a real investigational obesity drug with serious potential, but it is still a late-stage trial drug, not a normal public-use treatment.
Common mistakes
- Treating topline phase 3 headlines like they are the final complete answer.
- Comparing raw percentages across obesity-only and diabetes populations as if the populations were interchangeable.
- Assuming anything sold online as retatrutide is legitimate or safe because the molecule itself is in real clinical development.
- Ignoring older, approved options while waiting for an investigational medicine that is still not publicly available.
Clean test protocol
| Inputs | Your actual obesity or diabetes context, what approved options you have already considered, whether you meet trial-style risk criteria, and whether you are reading published data or just headlines |
|---|---|
| Duration | Reassess when full peer-reviewed phase 3 obesity publications arrive or when regulatory approval status changes. Until then, treat the current picture as promising but incomplete. |
| 3 metrics | Whether the weight-loss data stay strong in full publications, whether tolerability remains acceptable at high doses, and whether the drug clearly outperforms existing approved alternatives in the populations that matter most |
| Stop conditions | Any online seller or “research” product claiming to offer real retatrutide now, any blurred explanation of approval status, or any attempt to treat an investigational drug like a routine supplement decision |
How to tell it’s working
If retatrutide eventually becomes part of standard care, it should show sustained appetite reduction, clinically meaningful weight loss, and improvement in real weight-related health markers. Right now, for most readers, the more practical test is whether you can keep your view of the drug accurate instead of letting the headlines outrun the evidence.
Red flags / seek care
If you are taking any unverified product claimed to be retatrutide and develop persistent vomiting, dehydration, chest pain, severe palpitations, shortness of breath, or a strong allergic-type reaction, stop and seek medical care. Also treat fake-source risk as its own safety problem, not just a technicality.
Selected Professional References
These are the key sources behind the current retatrutide picture: mechanism, published phase 2 obesity data, newer phase 3 topline updates, approval status, and what is still underway.
What to Know About Retatrutide
The cleanest official source for what retatrutide is, where it is in development, and why it is not something the public should be using outside trials.
Used for: mechanism, approval status, trial-only availability, and current phase 3 scope
Triple-Hormone-Receptor Agonist Retatrutide for Obesity
Still the most important published obesity paper for retatrutide because it gives the strongest fully peer-reviewed view of the weight-loss and tolerability signal.
Used for: 24-week and 48-week weight loss, GI events, and heart-rate signal
Lilly Phase 2 Retatrutide Results Published in NEJM
Useful for the clean company summary of the phase 2 obesity readout and the “no plateau yet” detail that explains why interest accelerated so fast.
Used for: high-level phase 2 obesity context
TRIUMPH-4: Weight Loss and Knee Osteoarthritis Results
The strongest public phase 3 weight-loss readout so far, but still important to treat as topline until full publication arrives.
Used for: 28.7% at 68 weeks, pain improvement, and phase 3 tolerability context
TRANSCEND-T2D-1: First Phase 3 Diabetes Results
Important because it shows what retatrutide can do in type 2 diabetes, where weight loss is usually harder to achieve than in obesity-only populations.
Used for: 16.8% weight loss, A1C reduction, and GI AE rates in diabetes
TRIUMPH-5: Retatrutide Compared to Tirzepatide
This is one of the most important unanswered-trial pages because the head-to-head comparison will shape how people think about retatrutide’s real place in therapy.
Used for: why “better than tirzepatide?” is still unanswered
TRIUMPH-8: Retatrutide Weight-Loss Maintenance Trial
Maintenance is one of the most important real-world questions for any obesity drug, and this study exists because the answer is not assumed.
Used for: why durability and maintenance remain open questions
Cardiovascular Outcomes Study
A reminder that the deepest long-term cardiovascular questions are still being studied rather than assumed from early efficacy alone.
Used for: why long-term outcome confidence is still incomplete
Go Deeper (VerifiedSupps Guides)
These are better next reads if your real goal is metabolic health, smarter supplement choices, or protecting lean mass and health markers while managing weight.
How to Choose Supplements Without Guesswork
Best next read if you want a calmer framework before chasing shiny new therapies or headlines.
How Much Protein Do You Actually Need?
Helpful if your real question is how to protect lean mass and keep weight loss from becoming muscle loss.
Berberine Benefits
A better next comparison if your real focus is glucose and metabolic support rather than waiting on an investigational drug.
Omega-3 Complete Guide
Useful if you want to support cardiometabolic health with something more established while the retatrutide story continues to mature.
Final Takeaway
Retatrutide looks like a real contender, not a mirage. The weight-loss signal is already strong enough that it deserves serious attention. But the strongest published obesity evidence is still phase 2, the newest phase 3 wins are partly topline, and the biggest long-term questions are still in motion. The honest summary is not “it is overhyped,” and it is not “it is already settled.” It is “very promising, still investigational, and worth reading with real discipline.”
FAQ
Does retatrutide actually work for weight loss?
Yes. The current evidence strongly suggests that it does. The strongest published obesity dataset is still the phase 2 trial, while newer phase 3 public readouts look even stronger but are not yet all fully published in peer-reviewed form.
How does retatrutide work?
Retatrutide is a triple agonist that activates GIP, GLP-1, and glucagon receptors. The broader mechanism may help reduce food intake and, at least in preclinical work, increase energy expenditure.
How much weight loss has retatrutide shown so far?
In the published phase 2 obesity trial, the highest dose reached 24.2% average weight loss at 48 weeks. In the first successful public phase 3 obesity-plus-knee-osteoarthritis readout, the 12 mg dose reached 28.7% at 68 weeks. In the first phase 3 diabetes readout, the top dose reached 16.8% at 40 weeks.
Is retatrutide approved yet?
No. Retatrutide is still investigational and is not approved for public use.
Can you get retatrutide right now?
Legitimate access is currently through Lilly-sponsored clinical trials. It should not be treated like a routine publicly available medicine yet.
What side effects matter most with retatrutide?
The main side effects so far are gastrointestinal, including nausea, diarrhea, vomiting, and constipation, especially during dose escalation. Heart-rate increases and dysesthesia or hyperesthesia have also been reported.
Is retatrutide better than tirzepatide?
That is still an open question. A head-to-head obesity trial is ongoing, so the answer is not settled yet.
Why do some people treat retatrutide like a finished product already?
Because the early numbers are unusually strong and the mechanism is interesting. But strong early data are not the same thing as a fully mature approval, safety, and long-term outcomes package.
What still is not known about retatrutide?
Important unanswered questions include long-term cardiovascular and renal outcomes, durability of weight-loss maintenance, full peer-reviewed phase 3 obesity results, and direct comparison with tirzepatide.
What is the safest mindset right now?
Treat retatrutide as very promising but still incomplete. The calm view is stronger than either dismissal or hype.
VerifiedSupps Medical Disclaimer
This content is for educational purposes only and is not medical advice. Retatrutide is an investigational medicine, not an approved routine public-use therapy. It should not be treated as a substitute for medical evaluation, evidence-based obesity care, or approved treatment options. Be especially cautious with any product sold online as retatrutide outside a legitimate trial setting. Seek medical care for persistent vomiting, dehydration, chest pain, shortness of breath, severe palpitations, or any strong reaction after taking an unverified product.