By VerifiedSupps Editorial Team
Eloralintide for Weight Loss: Results, Side Effects, and Why Amylin Matters
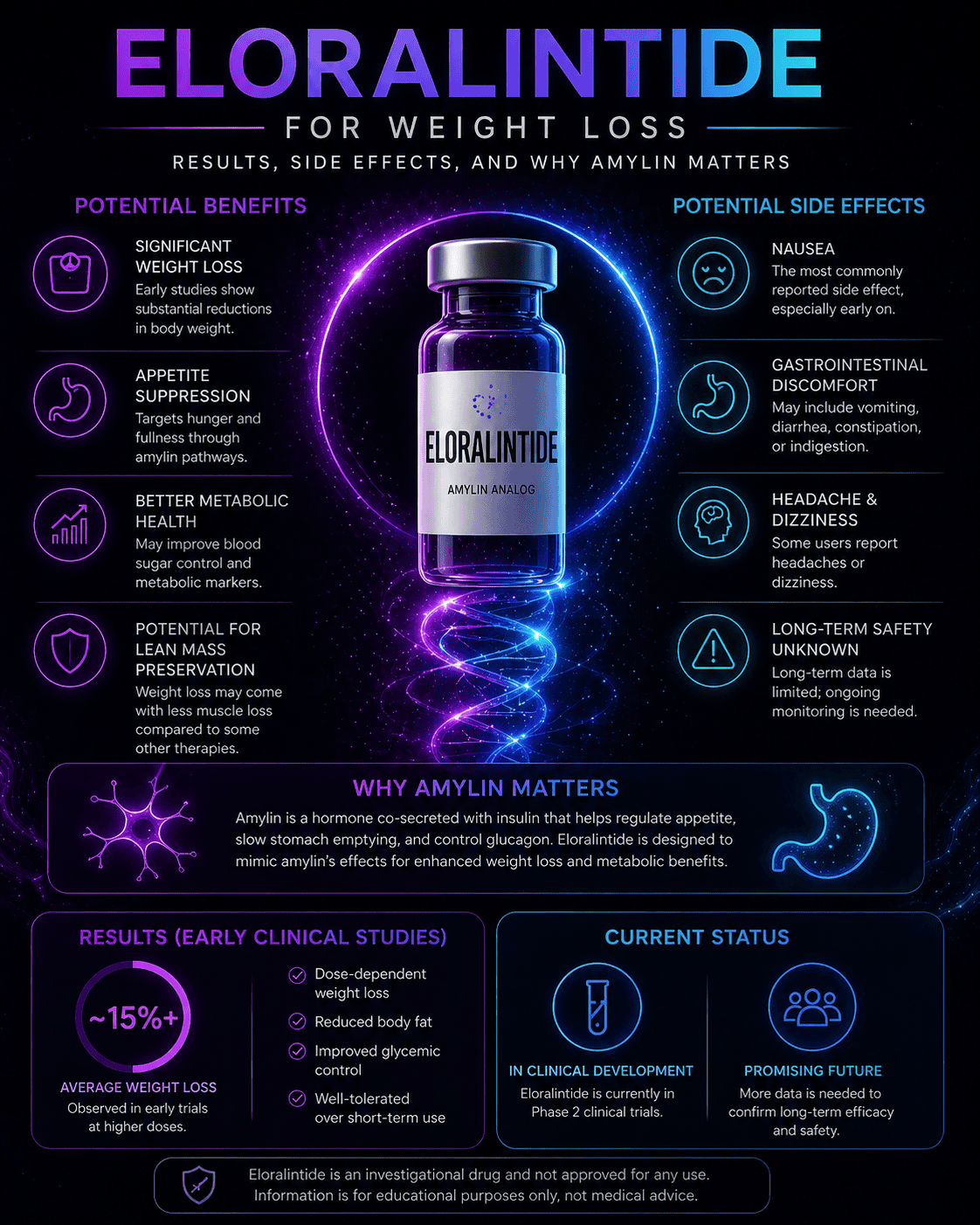
Eloralintide is an investigational once-weekly amylin receptor agonist being studied for obesity and weight management. The early signal is strong: a Phase 2 trial reported meaningful, dose-dependent weight loss over 48 weeks.
The practical takeaway is more cautious: eloralintide is promising, but it is not approved for routine use, and the best decisions still depend on full Phase 3 results, safety data, and medical supervision.
This guide focuses on eloralintide results, trial dosing, side effects, approval status, and the amylin mechanism; it does not provide injection instructions, sourcing advice, or treatment recommendations.
Key terms: eloralintide, LY3841136, amylin receptor agonist, AMY1R, obesity, weight management, tirzepatide combination research
Quick Take
Eloralintide appears to be one of the more important non-GLP-1 obesity-drug candidates to watch. Its Phase 2 data showed up to about 20% mean weight loss at 48 weeks, while nausea and fatigue were the main side-effect themes, especially at higher doses.
TL;DR decision
Follow eloralintide if you are tracking next-generation weight-loss medications, but do not treat it like an available supplement or approved prescription option. For weight-loss care today, use clinician-guided options and view eloralintide as an investigational therapy still moving through trials.
Evidence standard: human trials, dose ranges, guideline-level sources when available
Who this is for: readers comparing amylin-based weight-loss drugs, GLP-1 alternatives, and emerging obesity medication trial results
Who this is not for: anyone looking for self-injection instructions, research-vial sourcing, or a substitute for obesity care from a licensed clinician
Reviewed by: VerifiedSupps Editorial Team
Last reviewed: April 30, 2026
Parent Hub
VerifiedSupps Articles Library
Use the main article hub to compare evidence-based guides on peptides, obesity medications, metabolic health, and supplement safety in one place.
Does eloralintide work for weight loss?
Eloralintide appears to produce clinically meaningful weight loss in human trials, but it is still investigational. The strongest published signal so far is a 48-week Phase 2 trial showing dose-dependent weight reduction compared with placebo.
Mechanism
- Eloralintide targets amylin receptors, especially the amylin-1 receptor pathway being studied for satiety and food-intake control.
- Amylin is normally co-secreted with insulin after meals and helps signal fullness, slow gastric emptying, and regulate glucagon.
- The outcome is not just mechanism-driven; dose escalation, side-effect tolerance, diet quality, protein intake, activity, and trial duration all affect the real-world result.
Eloralintide decision decoder
Use this quick table to separate what eloralintide may mean from what it does not mean yet.
| What you want to know | Best read | What it means | Practical action |
|---|---|---|---|
| “How much weight loss did eloralintide show?” | About 9.5% to 20.1% mean weight loss across Phase 2 arms at 48 weeks | Strong Phase 2 signal, not final approval evidence | Watch Phase 3 outcomes and full safety reporting |
| “Is this another GLP-1?” | No, eloralintide is an amylin receptor agonist | It targets a different satiety pathway | Do not assume GLP-1 dosing, risks, or results apply directly |
| “Can I get it now?” | No approved routine-use product | This is trial and regulatory-review territory | Discuss approved medications or trial eligibility with a clinician |
| “What side effects matter most?” | Nausea and fatigue were the clearest Phase 2 themes | Tolerability may shape whether people can stay on therapy | Avoid unsupervised use and track safety data carefully |
Best next step (today): treat eloralintide as a promising investigational medication to monitor, not as something to buy, dose, or stack on your own.
What were the eloralintide trial results?
In the published Phase 2 obesity trial, eloralintide produced dose-dependent weight loss over 48 weeks in adults with obesity or overweight and at least one weight-related comorbidity, without type 2 diabetes. Placebo weight loss was small, which makes the active-treatment signal easier to interpret.
| Trial arm | Mean weight change at 48 weeks | Approximate kg / lb change | How to read it |
|---|---|---|---|
| Eloralintide 1 mg | -9.5% | -10.2 kg / -22.5 lb | Lowest studied dose still showed clinically meaningful loss |
| Eloralintide 3 mg | -12.4% | -13.3 kg / -29.3 lb | Moderate dose response with lower reported tolerability burden than higher arms |
| Eloralintide 6 mg | -17.6% | -18.7 kg / -41.2 lb | Large effect, but nausea was more frequent in this arm |
| Eloralintide 9 mg | -20.1% | -21.3 kg / -47.0 lb | Highest reported mean loss in the trial |
| Eloralintide 6/9 mg escalation | -19.9% | -21.0 kg / -46.3 lb | Suggests escalation may preserve efficacy while helping tolerability |
| Eloralintide 3/6/9 mg escalation | -16.4% | -17.8 kg / -39.2 lb | Strong loss with a more gradual dose pathway |
| Placebo | -0.4% | -0.2 kg / -0.4 lb | Small change compared with active treatment arms |
The result is impressive for Phase 2, but it should not be treated like a finished prescribing label. Trial design, dropout handling, side-effect rates, population differences, and Phase 3 replication all matter before firm comparisons can be made.
What dose of eloralintide is being studied?
There is no approved eloralintide dose for weight loss. In Phase 2, eloralintide was studied as a once-weekly subcutaneous injection at 1 mg, 3 mg, 6 mg, 9 mg, and escalation schedules reaching 9 mg.
| Dose context | What was studied | Why it matters |
|---|---|---|
| Fixed-dose arms | 1 mg, 3 mg, 6 mg, or 9 mg once weekly | Helped researchers identify dose-response patterns |
| Escalation arms | 6/9 mg and 3/6/9 mg escalation schedules | Escalation can influence tolerability and adherence |
| Phase 3 context | Registered studies are evaluating efficacy and safety in larger obesity populations | Late-stage trials help determine whether a dose is effective, tolerable, and approvable |
The key point is conservative: study doses are not self-use instructions. With injectable obesity medications, dose escalation, missed doses, medication interactions, gallbladder history, diabetes status, and GI tolerance can all change the safety picture.
How long does eloralintide take to work?
Eloralintide is not a “few days” weight-loss story. Phase 1 research reported meaningful short-term weight reduction after 12 weeks in early testing, while the main Phase 2 obesity data were measured over 48 weeks.
Appetite and fullness may change before major scale movement, but the clinically useful read is a trend over time: weight, waist, tolerability, nutrition quality, and whether the person can continue treatment safely.
Useful timing frame
For investigational medications, the fairest interpretation comes from trial endpoints and clinician-supervised monitoring, not from early anecdotes, social media claims, or one-week body-weight swings.
What side effects can eloralintide cause?
The main Phase 2 side-effect themes were nausea and fatigue, with gastrointestinal symptoms and fatigue appearing more often in higher-dose arms. Slower dose escalation appeared to reduce the incidence of these events, but complete Phase 3 safety data are still needed.
Most visible trial symptoms
Nausea and fatigue were the clearest reported patterns. These are not minor details; they can affect hydration, food tolerance, adherence, and whether a medication is sustainable.
What remains uncertain
Rare events, long-term tolerability, discontinuation patterns, and comparison against approved medications need larger and longer datasets.
| Side-effect theme | What Phase 2 suggests | Practical interpretation |
|---|---|---|
| Nausea | More common in several active-treatment arms, especially some higher-dose groups | Dose and escalation schedule may matter |
| Fatigue | Reported more often in higher-dose arms than placebo | Could affect training, workday energy, and adherence |
| General GI tolerance | Described as generally mild to moderate in the available Phase 2 reporting | Still needs larger Phase 3 confirmation before broad safety conclusions |
Anyone with significant digestive disease, gallbladder concerns, pancreatitis history, pregnancy considerations, diabetes medications, kidney or liver disease, or unexplained severe symptoms should treat this as medical territory.
Eloralintide troubleshooting: what if results or side effects do not make sense?
Because eloralintide is investigational, troubleshooting should not mean adjusting dose on your own. The right frame is to protect safety, avoid noisy interpretation, and use a clinical trial team or licensed clinician to interpret what is happening.
Common mistakes
- Treating Phase 2 results as if they were an approved label.
- Comparing eloralintide with GLP-1 drugs without matching trial duration, population, dose, and dropout handling.
- Ignoring basic weight-loss support: protein, resistance training, hydration, sleep, and enough micronutrient-dense food during appetite suppression.
Clean test protocol
| Inputs | Clinician-supervised medication plan or formal trial protocol, baseline weight, waist, blood pressure, relevant labs, medication list, side-effect history, protein target, hydration plan, and resistance-training baseline. |
|---|---|
| Duration | Use the trial protocol or clinician plan. For obesity medications in general, meaningful assessment usually requires weeks to months, not a few isolated weigh-ins. |
| 3 metrics | Weekly average weight trend, waist circumference trend, and side-effect burden with food tolerance and hydration notes. |
| Stop conditions | Stop self-adjusting and contact a clinician or study team for severe nausea, persistent vomiting, dehydration, fainting, severe abdominal pain, allergic symptoms, jaundice, chest pain, or any symptom that feels unsafe. |
How to tell it’s working
A good response is not just a lower scale weight. Better signs include steadier appetite, gradual waist reduction, manageable side effects, preserved strength where possible, and improving metabolic markers if those are being tracked.
Red flags / seek care
Seek medical help for persistent vomiting, severe or persistent abdominal pain, signs of dehydration, fainting, chest pain, yellowing skin or eyes, severe allergic symptoms, confusion, or thoughts of self-harm.
Why does amylin matter for weight loss?
Amylin matters because it gives researchers a different weight-loss pathway than GLP-1 alone. It is involved in fullness signaling, gastric emptying, and post-meal metabolic regulation, which makes it a logical target for appetite and weight management research.
Satiety
Amylin signaling may help people feel full with less food.
Meal flow
Amylin can influence gastric emptying and post-meal signals.
Combination logic
A separate pathway may pair with incretin-based therapy in future research.
That does not mean amylin drugs automatically beat GLP-1 or GIP/GLP-1 medications. It means researchers may have another tool for people who need different efficacy, tolerability, or combination strategies.
Is eloralintide approved or available now?
Eloralintide is not approved for routine clinical use. It has completed Phase 1 and Phase 2 work in adults with obesity or overweight without type 2 diabetes, and late-stage Phase 3 studies are now listed for larger obesity populations, including studies with and without type 2 diabetes.
Lilly is also studying eloralintide alone and in combination with tirzepatide. That combination idea is interesting because amylin and incretin pathways may complement each other, but it still needs controlled trial evidence before anyone should treat it as a practical treatment strategy.
Decision rule
Approved obesity medications belong in clinician-guided care. Eloralintide belongs in the “watch the trial data and regulatory pathway” category until approval, labeling, contraindications, and access rules are clear.
Selected Professional References
These sources support the trial results, mechanism explanation, investigational status, and safety framing used in this guide.
Eli Lilly Eloralintide Phase 2 Results Announcement
Supports the 48-week weight-loss range, dose arms, placebo comparison, common adverse events, and Phase 3 development plan.
Used for: trial results and status context
The Lancet Phase 2 Eloralintide Trial
Supports the 263-participant randomized Phase 2 study design, dose groups, 48-week endpoint, nausea and fatigue rates, and interpretation.
Used for: clinical efficacy and safety evidence
Lilly: What to Know About Eloralintide
Supports the investigational status, non-approval language, development phase, and monotherapy versus combination research context.
Used for: approval and development status
Lilly Medical: Selective Amylin Receptor Agonist
Supports the AMY1R selectivity explanation and the caution that the functional significance of selectivity in humans still requires further research.
Used for: receptor and mechanism framing
ClinicalTrials.gov: Eloralintide Without Type 2 Diabetes
Supports the current late-stage research context in adults with obesity or overweight who do not have type 2 diabetes.
Used for: current trial status context
ClinicalTrials.gov: Eloralintide With Type 2 Diabetes
Supports the current Phase 3 research context for adults with obesity or overweight and type 2 diabetes.
Used for: broader Phase 3 program context
Molecular Metabolism: Eloralintide Discovery to Proof of Concept
Supports the once-weekly pharmacokinetic rationale, early human tolerability findings, and AMY1R-selective mechanism context.
Used for: mechanism and early clinical evidence
Go Deeper (VerifiedSupps Guides)
Use these related guides to compare eloralintide with nearby investigational weight-loss medications and peptide safety topics without mixing up mechanisms, trial stages, and approved use.
Survodutide for Weight Loss
Compare another investigational obesity medication through results, side effects, and current status.
Petrelintide for Weight Loss
A useful companion guide for understanding another amylin-focused weight-loss candidate.
Amycretin for Weight Loss
Understand another amylin-linked obesity therapy and how early results should be interpreted.
Retatrutide: Weight Loss, Side Effects, and Status
Compare headline trial results across next-generation obesity medications without overreading early data.
Final Takeaway
Eloralintide is a promising investigational amylin-based weight-loss medication with strong Phase 2 results and a different mechanism than GLP-1 drugs. The best decision today is to watch the Phase 3 evidence and avoid unregulated access while using approved, clinician-guided options for real treatment decisions.
FAQ
Is eloralintide approved for weight loss?
No. Eloralintide is investigational and is not approved for routine clinical use. It is being evaluated in clinical trials for obesity and weight management.
How much weight loss did eloralintide show?
In a published Phase 2 trial, eloralintide showed mean weight loss from about 9.5% to 20.1% across active arms at 48 weeks, compared with about 0.4% with placebo.
Is eloralintide a GLP-1 drug?
No. Eloralintide is an amylin receptor agonist, not a GLP-1 receptor agonist. It targets a different satiety pathway than semaglutide-style GLP-1 medications.
What is amylin and why does it matter?
Amylin is a hormone co-secreted with insulin after meals. It helps regulate fullness, gastric emptying, and post-meal metabolic signals, which makes it a relevant target for weight-management research.
What are the main eloralintide side effects?
The clearest Phase 2 side-effect themes were nausea and fatigue, with gastrointestinal symptoms and fatigue more common in some higher-dose arms.
What dose of eloralintide was studied?
Phase 2 studied once-weekly eloralintide at 1 mg, 3 mg, 6 mg, 9 mg, and escalation schedules reaching 9 mg. These are study doses, not approved dosing instructions.
How long does eloralintide take to work?
The main Phase 2 obesity results were measured over 48 weeks. Early appetite or weight changes may occur sooner, but the clinically useful interpretation comes from longer supervised trends.
Is eloralintide better than Wegovy or Zepbound?
That is not proven. Eloralintide has promising Phase 2 data, but direct comparisons require matched populations, similar follow-up, complete safety data, and ideally head-to-head trials.
Is eloralintide being studied with tirzepatide?
Yes. Lilly is studying eloralintide both alone and in combination with tirzepatide. Combination use remains research territory until controlled data and regulatory decisions are available.
Can you buy eloralintide online?
There is no approved eloralintide product for routine use. Online research-vial listings are not the same as regulated clinical medication and should not be treated as a safe substitute.
VerifiedSupps Medical Disclaimer
Eloralintide is an investigational medication, not a dietary supplement. This article is for educational purposes only and does not provide medical advice, diagnosis, prescribing guidance, dosing instructions, or a recommendation to purchase unapproved products. Weight-loss medications and investigational therapies can cause meaningful side effects and may be unsafe with certain medical conditions or medications. Speak with a licensed clinician before considering any obesity medication, and seek urgent care for severe abdominal pain, persistent vomiting, dehydration, fainting, chest pain, jaundice, severe allergic symptoms, or thoughts of self-harm.