By VerifiedSupps Editorial Team
Retatrutide vs Tirzepatide: Which Is Better for Weight Loss Right Now?
If you mean the better real-world choice today, tirzepatide wins. It is approved, available, backed by large published phase 3 obesity data, and has much more mature labeling and long-term follow-up.
If you mean the molecule that may eventually push more pure weight loss, retatrutide is the more exciting contender. But that answer is still partly a phase-3-in-progress story, not a finished one. That is the main tension in this comparison.
This page is for the practical choice between retatrutide and tirzepatide: current weight-loss results, side-effect differences, approval status, and what is still genuinely unknown.
Key terms: retatrutide, tirzepatide, Zepbound, triple agonist, dual agonist, obesity medicine, weight loss, phase 3, current status
Quick Take
Choose tirzepatide if you want the better defined and accessible option today. Watch retatrutide if you care most about where obesity therapy may go next. The strongest current comparison is “tirzepatide wins on proof and availability; retatrutide may win later on pure efficacy, but that is not settled yet.”
TL;DR decision
Tirzepatide is the better choice right now. Retatrutide is the more speculative next-generation bet. That is the cleanest honest summary.
Evidence standard: human trials, dose ranges, guideline-level sources when available
Who this is for: people comparing obesity medicines and trying to understand whether retatrutide is actually better than tirzepatide or just newer and more exciting
Who this is not for: anyone looking for gray-market sourcing, trial shortcuts, or reassurance that an investigational medicine is safe to buy online
Reviewed by: VerifiedSupps Editorial Team
Last reviewed: April 17, 2026
Parent Hub
VerifiedSupps Articles
Use the broader article hub if you want a calmer framework for comparing new metabolic therapies without getting pulled around by headlines.
Quick decision table: which one fits your priority?
This is the fastest way to separate the better choice now from the more exciting possibility later.
| If your priority is… | Better pick today | Why | Confidence |
|---|---|---|---|
| The more proven obesity drug right now | Tirzepatide | Approved, labeled, published phase 3 obesity data, and long-term follow-up already exist | High |
| The molecule that may eventually push more pure weight loss | Retatrutide | Its strongest obesity signals are larger, but the evidence is less mature and not head-to-head yet | Moderate |
| Something you can legitimately access now | Tirzepatide | Retatrutide is still trial-only and not approved | High |
| A wait-and-see pipeline bet | Retatrutide | The head-to-head vs tirzepatide study is ongoing, so the upside case is still open | Moderate |
Best next step (today): If you need the better current choice, pick tirzepatide. If you are trying to forecast the next wave of obesity therapy, keep watching retatrutide.
Retatrutide vs tirzepatide: which is better right now?
Right now, tirzepatide is better. That is not because retatrutide looks weak. It is because tirzepatide already crossed the line from very promising to approved, labeled, and widely studied.
Retatrutide is the better “maybe later” answer if your question is which molecule may eventually push weight loss further. But that is still a forecast, not a finished conclusion. Today’s best answer and tomorrow’s most interesting answer are different here.
Mechanism
- Tirzepatide is a dual GIP and GLP-1 receptor agonist.
- Retatrutide is a triple agonist that adds glucagon-receptor activity to GIP and GLP-1 activity.
- That extra glucagon component is one reason retatrutide may eventually prove more potent for weight loss, but it also comes with a less mature tolerability and outcomes story.
What would change my recommendation: a published head-to-head result showing retatrutide clearly beats tirzepatide on clinically meaningful outcomes without unacceptable tolerability tradeoffs.
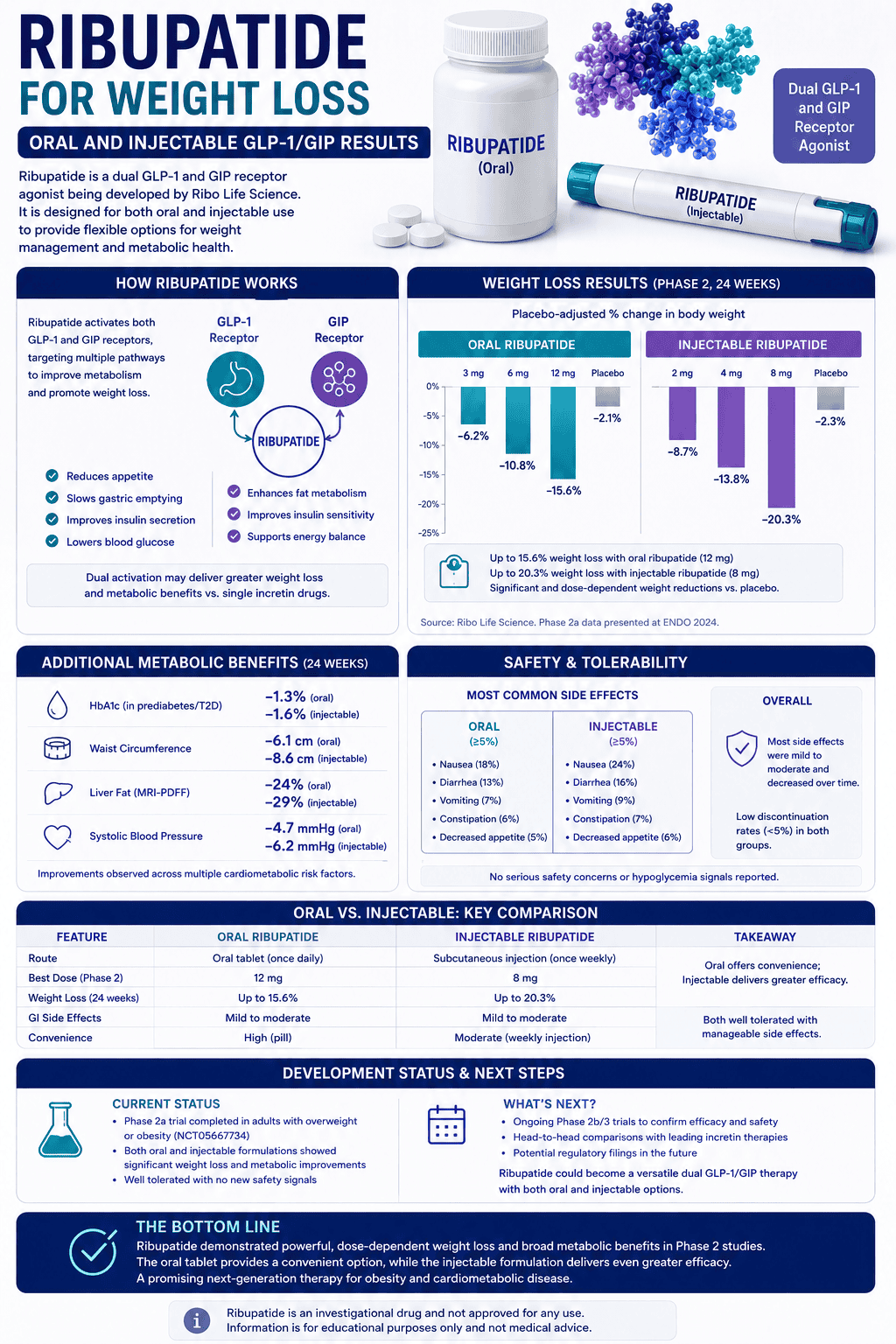
How much weight loss has retatrutide vs tirzepatide shown?
The simplest honest answer is that retatrutide looks stronger on the most eye-catching obesity numbers so far, but tirzepatide has the more mature and longer published evidence base. Those are not the same advantage.
Retatrutide
24.2%
Best published obesity result at 48 weeks in phase 2
The biggest public phase 3 number is even higher: 28.7% at 68 weeks in TRIUMPH-4, but that result is still topline rather than full publication.
Tirzepatide
22.5%
Efficacy-estimand weight loss at 72 weeks in SURMOUNT-1
It also has longer published follow-up: 22.9% at 176 weeks in the three-year SURMOUNT-1 prediabetes extension.
So which one is actually “stronger”? If you are just scanning top-line obesity numbers, retatrutide currently has the more aggressive public upside. If you care about what is most proven over time, tirzepatide still has the stronger real-world evidence stack.
Also keep the cross-trial problem in mind. These are different studies, different durations, and different populations. Until the direct comparison trial reads out, any numerical winner should be treated as provisional.
How do side effects compare between retatrutide and tirzepatide?
Both drugs live in the same broad tolerability neighborhood: gastrointestinal adverse effects are the main issue, especially during dose escalation. The more uncomfortable truth is that retatrutide may be harder on tolerability at the highest doses, but cross-trial comparisons still are not clean enough to call that final.
Tirzepatide today
Its common label-listed adverse reactions include nausea, diarrhea, vomiting, constipation, abdominal pain, dyspepsia, injection-site reactions, fatigue, hypersensitivity reactions, eructation, hair loss, and reflux.
In the weight-reduction program, the 15 mg arm reported nausea in 28%, diarrhea in 23%, vomiting in 13%, and discontinuation due to GI adverse reactions in 4.3%.
Retatrutide right now
The phase 3 toplines again show nausea, diarrhea, constipation, vomiting, and decreased appetite as the main problems, plus dysesthesia in a notable minority at higher doses.
In TRIUMPH-4, discontinuation due to adverse events reached 12.2% at 9 mg and 18.2% at 12 mg. That is a meaningful caution signal even if it is not a perfectly apples-to-apples comparison.
The clearest comparison today is this: tirzepatide has the more mature safety label, while retatrutide currently carries the more unsettled tolerability profile. That alone is enough to keep tirzepatide ahead in real-world decision-making.
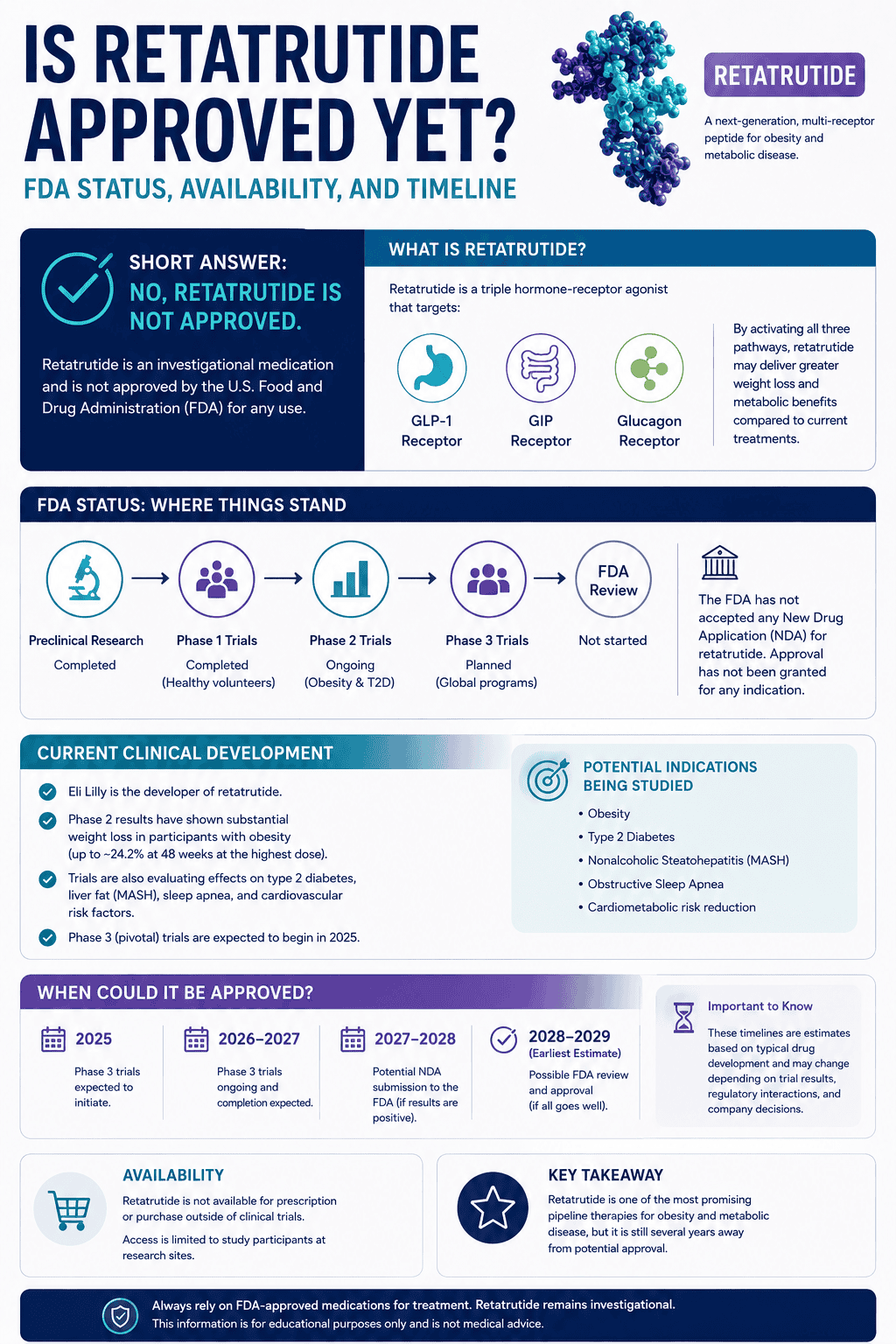
Which one is approved and available now?
Tirzepatide, clearly. Retatrutide is still trial-only.
Zepbound, the obesity brand of tirzepatide, is currently FDA-approved in combination with diet and physical activity to reduce excess body weight and maintain long-term weight reduction in adults with obesity or adults with overweight plus at least one weight-related comorbidity. It is also approved to treat moderate to severe obstructive sleep apnea in adults with obesity.
Retatrutide is different. Lilly says it is not approved, is legally available only to participants in Lilly clinical trials, and should not be used outside those trials. That difference alone settles the practical comparison for most people.
In other words, tirzepatide is a present-tense medical option. Retatrutide is still a future-tense one.
What should you choose if you are deciding between retatrutide and tirzepatide?
If the decision is for now, choose tirzepatide. If the decision is about what you want to keep watching, watch retatrutide. Most people do not need a more complicated answer than that.
Common mistakes
- Confusing a stronger-looking pipeline upside with a better current treatment choice.
- Comparing weight-loss percentages across unrelated studies as if that were the same as a head-to-head trial.
- Ignoring approval status and access as if they were minor details.
- Trusting anything sold online as retatrutide simply because the real molecule is in late-stage development.
Clean test protocol
| Inputs | Whether you need an approved therapy now, whether you care most about strongest current proof or highest future upside, and whether you are reading published evidence or just headlines |
|---|---|
| Duration | Reassess when the retatrutide-versus-tirzepatide head-to-head study reports and when more full retatrutide phase 3 obesity publications arrive |
| 3 metrics | Approval and access, published efficacy depth, and tolerability at clinically meaningful doses |
| Stop conditions | Any blurred explanation of approval status, any fake-source “research” product, or any decision based mainly on social-media enthusiasm instead of what is actually published |
How to tell it’s working
Right now, the more useful test is whether your framework is clear. A good comparison should leave you with the right answer for the right time horizon: tirzepatide for today, retatrutide as the more speculative future challenger.
Red flags / seek care
If you are using any unverified product claimed to be retatrutide or tirzepatide and develop persistent vomiting, dehydration, chest pain, severe palpitations, trouble breathing, or a strong allergic-type reaction, stop and seek medical care. Fake-source risk is part of the safety profile here.
Could retatrutide beat tirzepatide in the future?
Yes, it could. That is the whole reason this comparison matters. Retatrutide already has the kind of efficacy signals that make the question unavoidable.
But “could” is the right word. Retatrutide’s strongest current public obesity signal is bigger than tirzepatide’s best published obesity signal. At the same time, tirzepatide has the more mature evidence base, the more defined safety label, and real-world approval status. Those are different kinds of strength.
The head-to-head trial is what can finally turn this from an argument into an answer. Until then, the most disciplined view is that retatrutide may become the stronger pure efficacy drug, but tirzepatide remains the stronger current obesity medicine overall.
That is why the comparison is so interesting right now: it is one of the rare cases where the present winner and the possible future winner may not be the same drug.
Selected Professional References
These are the key sources behind the current comparison: published efficacy, topline signals, approved labeling, and the still-open head-to-head question.
Tirzepatide Once Weekly for the Treatment of Obesity
Still the anchor paper for tirzepatide’s original obesity case.
Used for: SURMOUNT-1 efficacy and early obesity tolerability
Tirzepatide Through 176 Weeks in SURMOUNT-1
Important because it gives tirzepatide something retatrutide does not have yet: more mature long-term obesity follow-up.
Used for: 22.9% at 176 weeks and 94% lower diabetes progression risk
Zepbound Prescribing Information
The most important source for tirzepatide’s current approved status, dosing, and adverse-reaction language.
Used for: approval status, warnings, and common side effects
Triple–Hormone-Receptor Agonist Retatrutide for Obesity
The anchor paper for the retatrutide weight-loss story in published form.
Used for: 48-week efficacy and phase 2 side-effect pattern
What to Know About Retatrutide
The cleanest official source for mechanism, current status, and trial-only availability.
Used for: triple-agonist mechanism and current status
TRIUMPH-4 Retatrutide Results
The biggest public retatrutide obesity number so far, with the important caveat that it is still topline.
Used for: 28.7% at 68 weeks and high-dose tolerability signal
TRANSCEND-T2D-1 Retatrutide Results
Useful because it shows the molecule is performing in more than one clinical setting and adds more current safety detail.
Used for: diabetes-phase efficacy and adverse-event rates
Retatrutide Compared With Tirzepatide in Adults With Obesity
This is the study that can eventually turn the comparison from informed forecast into a direct answer.
Used for: why the superiority question is still open
Go Deeper (VerifiedSupps Guides)
These are better next reads if you want the deeper retatrutide-specific picture or want to make smarter fat-loss decisions without relying on comparison headlines alone.
Retatrutide for Weight Loss
Best next read if you want the retatrutide-only efficacy story without the comparison layer.
Retatrutide: Results, Side Effects, and Current Status
Helpful if you want the cleaner retatrutide status snapshot after this head-to-head overview.
How Much Protein Do You Actually Need?
Useful if your real concern is lean-mass protection while weight is coming down.
How to Choose Supplements Without Guesswork
The best next read if this comparison mainly reminded you how easy it is to confuse excitement with certainty.
Final Takeaway
Tirzepatide is the better obesity medicine right now because it is approved, available, and supported by more mature efficacy and safety data. Retatrutide is the more intriguing future challenger because its strongest current weight-loss signals are even more aggressive. Until the direct comparison study reads out, the cleanest answer is not “retatrutide beats tirzepatide” or “tirzepatide has nothing to worry about.” It is “tirzepatide wins today, retatrutide may push the field further tomorrow.”
FAQ
Which is better right now, retatrutide or tirzepatide?
Right now, tirzepatide is the better choice because it is approved, available, and supported by more mature published obesity data and labeling.
Could retatrutide end up stronger than tirzepatide?
Yes, it could. Retatrutide already has very strong weight-loss signals, but the direct head-to-head answer is still not available.
How much weight loss has retatrutide shown so far?
The best published obesity result so far is 24.2% average weight loss at 48 weeks in phase 2, and the biggest public phase 3 number is 28.7% at 68 weeks in TRIUMPH-4.
How much weight loss has tirzepatide shown so far?
In SURMOUNT-1, tirzepatide reached up to 22.5% average weight loss at 72 weeks using the efficacy estimand, and it reached 22.9% at 176 weeks with the 15 mg dose in the three-year prediabetes follow-up.
Which has more mature safety information?
Tirzepatide does. It has an FDA-approved prescribing label with established warnings, common adverse reactions, and approved-dose guidance.
What side effects matter most in both drugs?
Gastrointestinal side effects matter most in both, especially nausea, diarrhea, vomiting, and constipation during dose escalation.
Is retatrutide approved yet?
No. Retatrutide is still investigational and is legally available only in Lilly-sponsored clinical trials.
Is tirzepatide approved for obesity?
Yes. Zepbound is FDA-approved for chronic weight management in adults with obesity or adults with overweight plus at least one weight-related comorbidity, and it is also approved for moderate to severe obstructive sleep apnea in adults with obesity.
Is there a direct retatrutide vs tirzepatide study underway?
Yes. A head-to-head study in adults with obesity is underway, so the direct superiority question is still open.
What is the safest current mindset?
Treat tirzepatide as the better current option and retatrutide as the more speculative next-generation contender until the head-to-head and fuller phase 3 evidence arrive.
VerifiedSupps Medical Disclaimer
This content is for educational purposes only and is not medical advice. Tirzepatide is an FDA-approved prescription medicine with labeled indications, warnings, and dose-escalation guidance. Retatrutide is still investigational and should not be treated as a normal public-use option. Do not use unverified products sold as either medicine as a substitute for medical care, approved treatment, or legitimate clinical-trial participation. Seek urgent medical care for severe vomiting, dehydration, chest pain, shortness of breath, severe palpitations, or a strong allergic-type reaction after using any unverified injectable product.